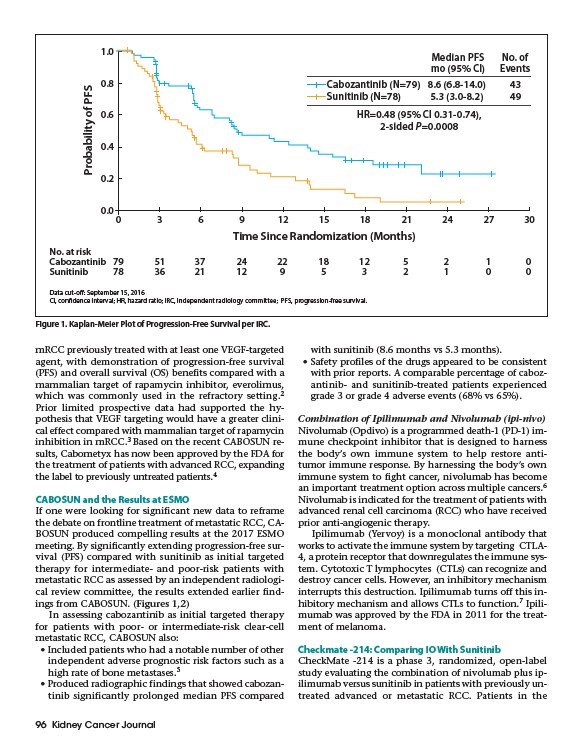
1.0
0.8
0.6
0.4
0.2
0.0
Median PFS
mo (95% CI)
0 3 6 9 12 15 18 21 24 27 30
Time Since Randomization (Months)
Probability of PFS
No. at risk
Cabozantinib
Sunitinib
79 51 37 24 22 18 12 5 2 1 0
78 36 21 12 9 5 3 2 1 0 0
Data cut-off: September 15, 2016
CI, confidence interval; HR, hazard ratio; IRC, independent radiology committee; PFS, progression-free survival.
mRCC previously treated with at least one VEGF-targeted
agent, with demonstration of progression-free survival
(PFS) and overall survival (OS) benefits compared with a
mammalian target of rapamycin inhibitor, everolimus,
which was commonly used in the refractory setting.2
Prior limited prospective data had supported the hypothesis
that VEGF targeting would have a greater clinical
effect compared with mammalian target of rapamycin
inhibition in mRCC.3 Based on the recent CABOSUN results,
Cabometyx has now been approved by the FDA for
the treatment of patients with advanced RCC, expanding
the label to previously untreated patients.4
CABOSUN and the Results at ESMO
If one were looking for significant new data to reframe
the debate on frontline treatment of metastatic RCC, CABOSUN
produced compelling results at the 2017 ESMO
meeting. By significantly extending progression-free survival
(PFS) compared with sunitinib as initial targeted
therapy for intermediate- and poor-risk patients with
metastatic RCC as assessed by an independent radiological
review committee, the results extended earlier findings
from CABOSUN. (Figures 1,2)
In assessing cabozantinib as initial targeted therapy
for patients with poor- or intermediate-risk clear-cell
metastatic RCC, CABOSUN also:
• Included patients who had a notable number of other
independent adverse prognostic risk factors such as a
high rate of bone metastases.5
• Produced radiographic findings that showed cabozantinib
significantly prolonged median PFS compared
96 Kidney Cancer Journal
with sunitinib (8.6 months vs 5.3 months).
• Safety profiles of the drugs appeared to be consistent
with prior reports. A comparable percentage of cabozantinib
and sunitinib-treated patients experienced
grade 3 or grade 4 adverse events (68% vs 65%).
Combination of Ipilimumab and Nivolumab (ipi-nivo)
Nivolumab (Opdivo) is a programmed death-1 (PD-1) immune
checkpoint inhibitor that is designed to harness
the body’s own immune system to help restore antitumor
immune response. By harnessing the body’s own
immune system to fight cancer, nivolumab has become
an important treatment option across multiple cancers.6
Nivolumab is indicated for the treatment of patients with
advanced renal cell carcinoma (RCC) who have received
prior anti-angiogenic therapy.
Ipilimumab (Yervoy) is a monoclonal antibody that
works to activate the immune system by targeting CTLA-
4, a protein receptor that downregulates the immune system.
Cytotoxic T lymphocytes (CTLs) can recognize and
destroy cancer cells. However, an inhibitory mechanism
interrupts this destruction. Ipilimumab turns off this inhibitory
mechanism and allows CTLs to function.7 Ipilimumab
was approved by the FDA in 2011 for the treatment
of melanoma.
Checkmate -214: Comparing IO With Sunitinib
CheckMate -214 is a phase 3, randomized, open-label
study evaluating the combination of nivolumab plus ipilimumab
versus sunitinib in patients with previously untreated
advanced or metastatic RCC. Patients in the
Figure 1. Kaplan-Meier Plot of Progression-Free Survival per IRC.
Cabozantinib (N=79) 8.6 (6.8-14.0) 43
Sunitinib (N=78) 5.3 (3.0-8.2) 49
HR=0.48 (95% CI 0.31-0.74),
2-sided P=0.0008
No. of
Events