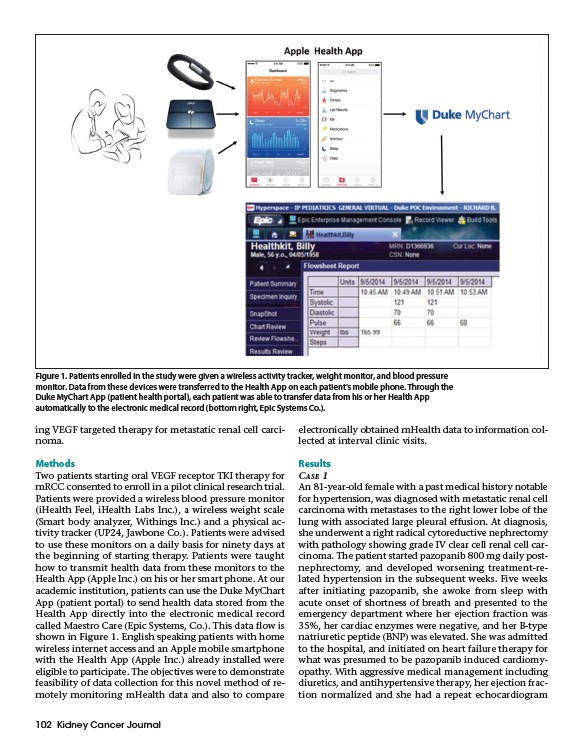
Figure 1. Patients enrolled in the study were given a wireless activity tracker, weight monitor, and blood pressure
monitor. Data from these devices were transferred to the Health App on each patient’s mobile phone. Through the
Duke MyChart App (patient health portal), each patient was able to transfer data from his or her Health App
automatically to the electronic medical record (bottom right, Epic Systems Co.).
ing VEGF targeted therapy for metastatic renal cell carcinoma.
Methods
Two patients starting oral VEGF receptor TKI therapy for
mRCC consented to enroll in a pilot clinical research trial.
Patients were provided a wireless blood pressure monitor
(iHealth Feel, iHealth Labs Inc.), a wireless weight scale
(Smart body analyzer, Withings Inc.) and a physical activity
tracker (UP24, Jawbone Co.). Patients were advised
to use these monitors on a daily basis for ninety days at
the beginning of starting therapy. Patients were taught
how to transmit health data from these monitors to the
Health App (Apple Inc.) on his or her smart phone. At our
academic institution, patients can use the Duke MyChart
App (patient portal) to send health data stored from the
Health App directly into the electronic medical record
called Maestro Care (Epic Systems, Co.). This data flow is
shown in Figure 1. English speaking patients with home
wireless internet access and an Apple mobile smartphone
with the Health App (Apple Inc.) already installed were
eligible to participate. The objectives were to demonstrate
feasibility of data collection for this novel method of remotely
monitoring mHealth data and also to compare
102 Kidney Cancer Journal
electronically obtained mHealth data to information collected
at interval clinic visits.
Results
CASE 1
An 81-year-old female with a past medical history notable
for hypertension, was diagnosed with metastatic renal cell
carcinoma with metastases to the right lower lobe of the
lung with associated large pleural effusion. At diagnosis,
she underwent a right radical cytoreductive nephrectomy
with pathology showing grade IV clear cell renal cell carcinoma.
The patient started pazopanib 800 mg daily postnephrectomy,
and developed worsening treatment-related
hypertension in the subsequent weeks. Five weeks
after initiating pazopanib, she awoke from sleep with
acute onset of shortness of breath and presented to the
emergency department where her ejection fraction was
35%, her cardiac enzymes were negative, and her B-type
natriuretic peptide (BNP) was elevated. She was admitted
to the hospital, and initiated on heart failure therapy for
what was presumed to be pazopanib induced cardiomyopathy.
With aggressive medical management including
diuretics, and antihypertensive therapy, her ejection fraction
normalized and she had a repeat echocardiogram