3. During ascent, the reduction of pressure will cause
the gas in our lungs to expand. If you hold your
breath, not allowing the excess gas to be exhaled,
you risk rupturing your lungs–with possibly
calamitous consequences.
You may also remember the other gas laws that
relate temperature to volume (if the pressure is held constant)
and temperature to pressure (if the volume is held
constant). These have less obvious relevance in enriched
air nitrox diving, but you will get to know them well if
you take the NAUI Master Scuba Diver course.
HENRY’S LAW: THE SOLUBILITY OF GASES
Gases dissolve in liquids. Solids dissolve in liquids
too. You know that sugar dissolves in water because you
can see it happen. Most persons are less aware that gases
also dissolve, although carbonated beverages are a perfect
everyday example of gas solubility. Some gases are
more soluble in a liquid than other gases, and some liquids
are better solvents of a gas than other liquids.
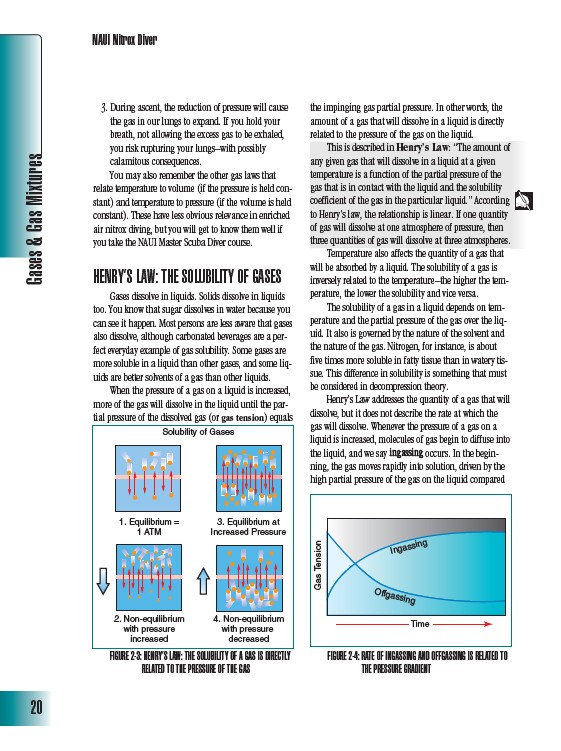
When the pressure of a gas on a liquid is increased,
more of the gas will dissolve in the liquid until the partial
pressure of the dissolved gas (or gas tension) equals
the impinging gas partial pressure. In other words, the
amount of a gas that will dissolve in a liquid is directly
related to the pressure of the gas on the liquid.
This is described in Henry’s Law: “The amount of
any given gas that will dissolve in a liquid at a given
temperature is a function of the partial pressure of the
gas that is in contact with the liquid and the solubility
coefficient of the gas in the particular liquid.” According
to Henry’s law, the relationship is linear. If one quantity
of gas will dissolve at one atmosphere of pressure, then
three quantities of gas will dissolve at three atmospheres.
Temperature also affects the quantity of a gas that
will be absorbed by a liquid. The solubility of a gas is
inversely related to the temperature–the higher the temperature,
the lower the solubility and vice versa.
The solubility of a gas in a liquid depends on temperature
and the partial pressure of the gas over the liquid.
It also is governed by the nature of the solvent and
the nature of the gas. Nitrogen, for instance, is about
five times more soluble in fatty tissue than in watery tissue.
This difference in solubility is something that must
be considered in decompression theory.
Henry’s Law addresses the quantity of a gas that will
dissolve, but it does not describe the rate at which the
gas will dissolve. Whenever the pressure of a gas on a
liquid is increased, molecules of gas begin to diffuse into
the liquid, and we say ingassing occurs. In the beginning,
the gas moves rapidly into solution, driven by the
high partial pressure of the gas on the liquid compared
NAUI Nitrox Diver
20 Gases & Gas Mixtures
FIGURE 2-4: RATE OF INGASSING AND OFFGASSING IS RELATED TO
THE PRESSURE GRADIENT
Solubility of Gases
1. Equilibrium =
1 ATM
2. Non-equilibrium
with pressure
increased
3. Equilibrium at
Increased Pressure
4. Non-equilibrium
with pressure
decreased
FIGURE 2-3: HENRY’S LAW: THE SOLUBILITY OF A GAS IS DIRECTLY
RELATED TO THE PRESSURE OF THE GAS
Ingassing
Offgassing
Gas Tension
Time