Kidney Cancer Journal 23
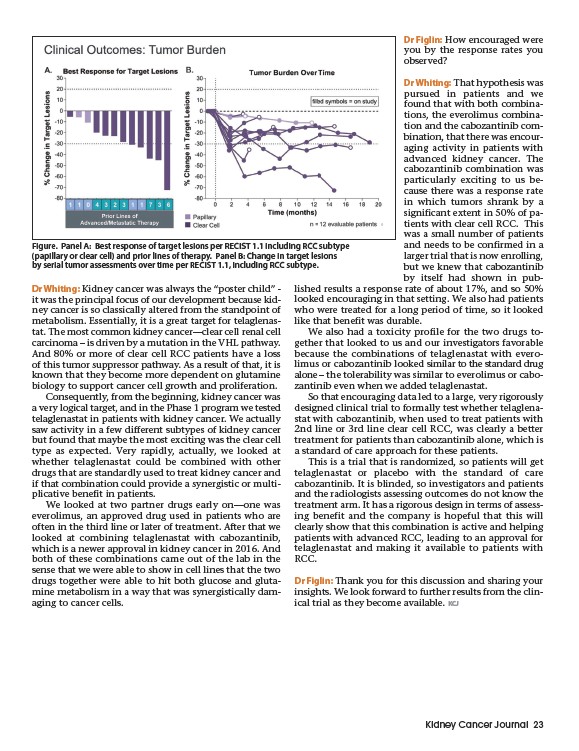
Figure. Panel A: Best response of target lesions per RECIST 1.1 including RCC subtype
(papillary or clear cell) and prior lines of therapy. Panel B: Change in target lesions
by serial tumor assessments over time per RECIST 1.1, including RCC subtype.
Dr Whiting: Kidney cancer was always the “poster child” -
it was the principal focus of our development because kidney
cancer is so classically altered from the standpoint of
metabolism. Essentially, it is a great target for telaglenastat.
The most common kidney cancer—clear cell renal cell
carcinoma – is driven by a mutation in the VHL pathway.
And 80% or more of clear cell RCC patients have a loss
of this tumor suppressor pathway. As a result of that, it is
known that they become more dependent on glutamine
biology to support cancer cell growth and proliferation.
Consequently, from the beginning, kidney cancer was
a very logical target, and in the Phase 1 program we tested
telaglenastat in patients with kidney cancer. We actually
saw activity in a few different subtypes of kidney cancer
but found that maybe the most exciting was the clear cell
type as expected. Very rapidly, actually, we looked at
whether telaglenastat could be combined with other
drugs that are standardly used to treat kidney cancer and
if that combination could provide a synergistic or multiplicative
benefit in patients.
We looked at two partner drugs early on—one was
everolimus, an approved drug used in patients who are
often in the third line or later of treatment. After that we
looked at combining telaglenastat with cabozantinib,
which is a newer approval in kidney cancer in 2016. And
both of these combinations came out of the lab in the
sense that we were able to show in cell lines that the two
drugs together were able to hit both glucose and glutamine
metabolism in a way that was synergistically damaging
to cancer cells.
Dr Figlin: How encouraged were
you by the response rates you
observed?
Dr Whiting: That hypothesis was
pursued in patients and we
found that with both combinations,
the everolimus combination
and the cabozantinib com-
bination, that there was encouraging
activity in patients with
advanced kidney cancer. The
cabozantinib combination was
particularly exciting to us because
there was a response rate
in which tumors shrank by a
significant extent in 50% of patients
with clear cell RCC. This
was a small number of patients
and needs to be confirmed in a
larger trial that is now enrolling,
but we knew that cabozantinib
by itself had shown in published
results a response rate of about 17%, and so 50%
looked encouraging in that setting. We also had patients
who were treated for a long period of time, so it looked
like that benefit was durable.
We also had a toxicity profile for the two drugs together
that looked to us and our investigators favorable
because the combinations of telaglenastat with everolimus
or cabozantinib looked similar to the standard drug
alone – the tolerability was similar to everolimus or cabozantinib
even when we added telaglenastat.
So that encouraging data led to a large, very rigorously
designed clinical trial to formally test whether telaglenastat
with cabozantinib, when used to treat patients with
2nd line or 3rd line clear cell RCC, was clearly a better
treatment for patients than cabozantinib alone, which is
a standard of care approach for these patients.
This is a trial that is randomized, so patients will get
telaglenastat or placebo with the standard of care
cabozantinib. It is blinded, so investigators and patients
and the radiologists assessing outcomes do not know the
treatment arm. It has a rigorous design in terms of assessing
benefit and the company is hopeful that this will
clearly show that this combination is active and helping
patients with advanced RCC, leading to an approval for
telaglenastat and making it available to patients with
RCC.
Dr Figlin: Thank you for this discussion and sharing your
insights. We look forward to further results from the clinical
trial as they become available. KCJ