Kidney Cancer Journal 71
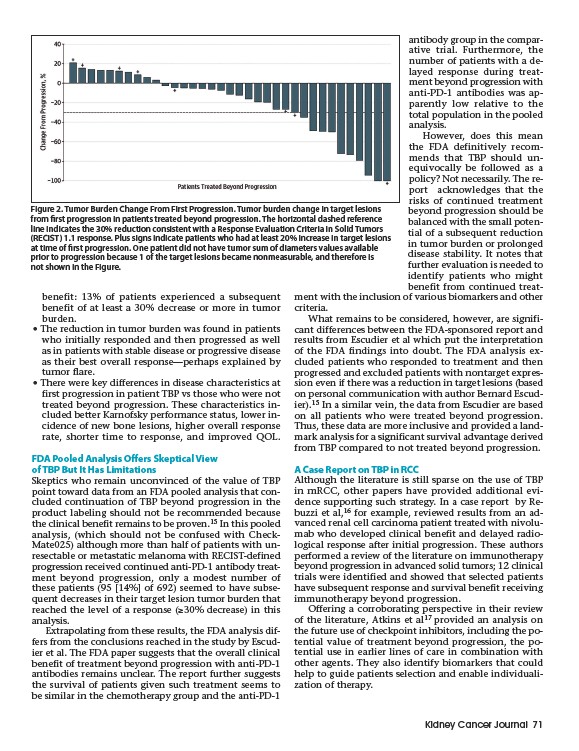
Figure 2. Tumor Burden Change From First Progression. Tumor burden change in target lesions
from first progression in patients treated beyond progression. The horizontal dashed reference
line indicates the 30% reduction consistent with a Response Evaluation Criteria in Solid Tumors
(RECIST) 1.1 response. Plus signs indicate patients who had at least 20% increase in target lesions
at time of first progression. One patient did not have tumor sum of diameters values available
prior to progression because 1 of the target lesions became nonmeasurable, and therefore is
not shown in the Figure.
benefit: 13% of patients experienced a subsequent
benefit of at least a 30% decrease or more in tumor
burden.
• The reduction in tumor burden was found in patients
who initially responded and then progressed as well
as in patients with stable disease or progressive disease
as their best overall response—perhaps explained by
tumor flare.
• There were key differences in disease characteristics at
first progression in patient TBP vs those who were not
treated beyond progression. These characteristics included
better Karnofsky performance status, lower incidence
of new bone lesions, higher overall response
rate, shorter time to response, and improved QOL.
FDA Pooled Analysis Offers Skeptical View
of TBP But It Has Limitations
Skeptics who remain unconvinced of the value of TBP
point toward data from an FDA pooled analysis that concluded
continuation of TBP beyond progression in the
product labeling should not be recommended because
the clinical benefit remains to be proven.15 In this pooled
analysis, (which should not be confused with Check-
Mate025) although more than half of patients with unresectable
or metastatic melanoma with RECIST-defined
progression received continued anti-PD-1 antibody treatment
beyond progression, only a modest number of
these patients (95 14% of 692) seemed to have subsequent
decreases in their target lesion tumor burden that
reached the level of a response (≥30% decrease) in this
analysis.
Extrapolating from these results, the FDA analysis differs
from the conclusions reached in the study by Escudier
et al. The FDA paper suggests that the overall clinical
benefit of treatment beyond progression with anti-PD-1
antibodies remains unclear. The report further suggests
the survival of patients given such treatment seems to
be similar in the chemotherapy group and the anti-PD-1
antibody group in the comparative
trial. Furthermore, the
number of patients with a delayed
response during treatment
beyond progression with
anti-PD-1 antibodies was apparently
low relative to the
total population in the pooled
analysis.
However, does this mean
the FDA definitively recommends
that TBP should unequivocally
be followed as a
policy? Not necessarily. The report
acknowledges that the
risks of continued treatment
beyond progression should be
balanced with the small potential
of a subsequent reduction
in tumor burden or prolonged
disease stability. It notes that
further evaluation is needed to
identify patients who might
benefit from continued treatment
with the inclusion of various biomarkers and other
criteria.
What remains to be considered, however, are significant
differences between the FDA-sponsored report and
results from Escudier et al which put the interpretation
of the FDA findings into doubt. The FDA analysis excluded
patients who responded to treatment and then
progressed and excluded patients with nontarget expression
even if there was a reduction in target lesions (based
on personal communication with author Bernard Escudier).
15 In a similar vein, the data from Escudier are based
on all patients who were treated beyond progression.
Thus, these data are more inclusive and provided a landmark
analysis for a significant survival advantage derived
from TBP compared to not treated beyond progression.
A Case Report on TBP in RCC
Although the literature is still sparse on the use of TBP
in mRCC, other papers have provided additional evidence
supporting such strategy. In a case report by Rebuzzi
et al,16 for example, reviewed results from an ad-
vanced renal cell carcinoma patient treated with nivolumab
who developed clinical benefit and delayed radiological
response after initial progression. These authors
performed a review of the literature on immunotherapy
beyond progression in advanced solid tumors; 12 clinical
trials were identified and showed that selected patients
have subsequent response and survival benefit receiving
immunotherapy beyond progression.
Offering a corroborating perspective in their review
of the literature, Atkins et al17 provided an analysis on
the future use of checkpoint inhibitors, including the potential
value of treatment beyond progression, the potential
use in earlier lines of care in combination with
other agents. They also identify biomarkers that could
help to guide patients selection and enable individualization
of therapy.