Kidney Cancer Journal 75
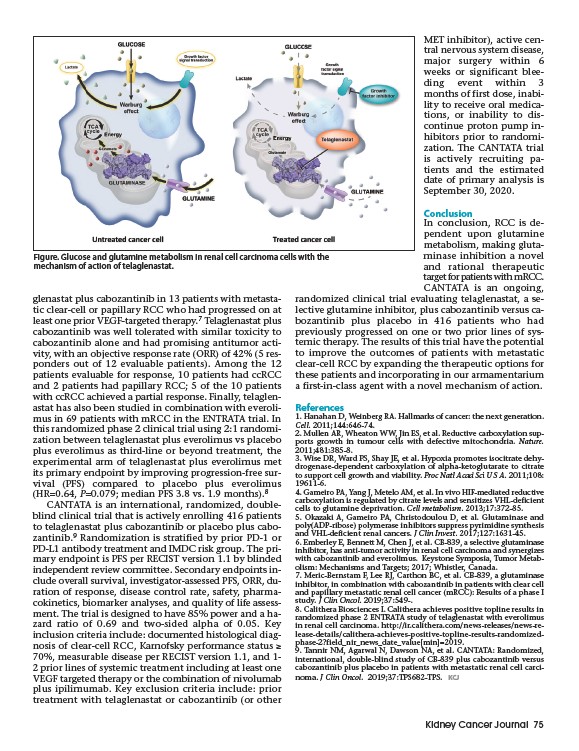
Figure. Glucose and glutamine metabolism in renal cell carcinoma cells with the
mechanism of action of telaglenastat.
glenastat plus cabozantinib in 13 patients with metastatic
clear-cell or papillary RCC who had progressed on at
least one prior VEGF-targeted therapy.7 Telaglenastat plus
cabozantinib was well tolerated with similar toxicity to
cabozantinib alone and had promising antitumor activity,
with an objective response rate (ORR) of 42% (5 responders
out of 12 evaluable patients). Among the 12
patients evaluable for response, 10 patients had ccRCC
and 2 patients had papillary RCC; 5 of the 10 patients
with ccRCC achieved a partial response. Finally, telaglenastat
has also been studied in combination with everolimus
in 69 patients with mRCC in the ENTRATA trial. In
this randomized phase 2 clinical trial using 2:1 randomization
between telaglenastat plus everolimus vs placebo
plus everolimus as third-line or beyond treatment, the
experimental arm of telaglenastat plus everolimus met
its primary endpoint by improving progression-free survival
(PFS) compared to placebo plus everolimus
(HR=0.64, P=0.079; median PFS 3.8 vs. 1.9 months).8
CANTATA is an international, randomized, doubleblind
clinical trial that is actively enrolling 416 patients
to telaglenastat plus cabozantinib or placebo plus cabozantinib.
9 Randomization is stratified by prior PD-1 or
PD-L1 antibody treatment and IMDC risk group. The primary
endpoint is PFS per RECIST version 1.1 by blinded
independent review committee. Secondary endpoints include
overall survival, investigator-assessed PFS, ORR, duration
of response, disease control rate, safety, pharma-
cokinetics, biomarker analyses, and quality of life assessment.
The trial is designed to have 85% power and a hazard
ratio of 0.69 and two-sided alpha of 0.05. Key
inclusion criteria include: documented histological diagnosis
of clear-cell RCC, Karnofsky performance status ≥
70%, measurable disease per RECIST version 1.1, and 1-
2 prior lines of systemic treatment including at least one
VEGF targeted therapy or the combination of nivolumab
plus ipilimumab. Key exclusion criteria include: prior
treatment with telaglenastat or cabozantinib (or other
MET inhibitor), active central
nervous system disease,
major surgery within 6
weeks or significant bleeding
event within 3
months of first dose, inability
to receive oral medications,
or inability to dis-
continue proton pump inhibitors
prior to randomization.
The CANTATA trial
is actively recruiting patients
and the estimated
date of primary analysis is
September 30, 2020.
Conclusion
In conclusion, RCC is dependent
upon glutamine
metabolism, making glutaminase
inhibition a novel
and rational therapeutic
target for patients with mRCC.
CANTATA is an ongoing,
randomized clinical trial evaluating telaglenastat, a selective
glutamine inhibitor, plus cabozantinib versus cabozantinib
plus placebo in 416 patients who had
previously progressed on one or two prior lines of systemic
therapy. The results of this trial have the potential
to improve the outcomes of patients with metastatic
clear-cell RCC by expanding the therapeutic options for
these patients and incorporating in our armamentarium
a first-in-class agent with a novel mechanism of action.
References
1. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation.
Cell. 2011;144:646-74.
2. Mullen AR, Wheaton WW, Jin ES, et al. Reductive carboxylation supports
growth in tumour cells with defective mitochondria. Nature.
2011;481:385-8.
3. Wise DR, Ward PS, Shay JE, et al. Hypoxia promotes isocitrate dehydrogenase
dependent carboxylation of alpha-ketoglutarate to citrate
to support cell growth and viability. Proc Natl Acad Sci U S A. 2011;108:
19611-6.
4. Gameiro PA, Yang J, Metelo AM, et al. In vivo HIF-mediated reductive
carboxylation is regulated by citrate levels and sensitizes VHL-deficient
cells to glutamine deprivation. Cell metabolism. 2013;17:372-85.
5. Okazaki A, Gameiro PA, Christodoulou D, et al. Glutaminase and
poly(ADP-ribose) polymerase inhibitors suppress pyrimidine synthesis
and VHL-deficient renal cancers. J Clin Invest. 2017;127:1631-45.
6. Emberley E, Bennett M, Chen J, et al. CB-839, a selective glutaminase
inhibitor, has anti-tumor activity in renal cell carcinoma and synergizes
with cabozantinib and everolimus. Keystone Symposia, Tumor Metabolism:
Mechanisms and Targets; 2017; Whistler, Canada.
7. Meric-Bernstam F, Lee RJ, Carthon BC, et al. CB-839, a glutaminase
inhibitor, in combination with cabozantinib in patients with clear cell
and papillary metastatic renal cell cancer (mRCC): Results of a phase I
study. J Clin Oncol. 2019;37:549-.
8. Calithera Biosciences I. Calithera achieves positive topline results in
randomized phase 2 ENTRATA study of telaglenastat with everolimus
in renal cell carcinoma. http://ir.calithera.com/news-releases/news-release
details/calithera-achieves-positive-topline-results-randomizedphase
2?field_nir_news_date_valuemin=2019.
9. Tannir NM, Agarwal N, Dawson NA, et al. CANTATA: Randomized,
international, double-blind study of CB-839 plus cabozantinib versus
cabozantinib plus placebo in patients with metastatic renal cell carcinoma.
J Clin Oncol. 2019;37:TPS682-TPS. KCJ