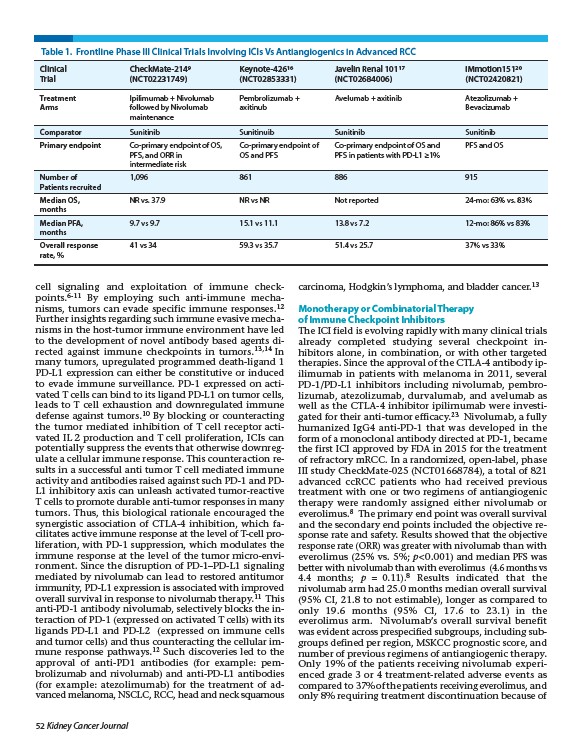
Table 1. Frontline Phase III Clinical Trials Involving ICIs Vs Antiangiogenics in Advanced RCC
Clinical CheckMate-2149 Keynote-42616 Javelin Renal 10117 IMmotion15120
Trial (NCT02231749) (NCT02853331) (NCT02684006) (NCT02420821)
Treatment Ipilimumab + Nivolumab Pembrolizumab + Avelumab + axitinib Atezolizumab +
Arms followed by Nivolumab axitinub Bevacizumab
cell signaling and exploitation of immune checkpoints.
6-11 By employing such anti-immune mechanisms,
tumors can evade specific immune responses.12
Further insights regarding such immune evasive mechanisms
in the host-tumor immune environment have led
to the development of novel antibody based agents directed
against immune checkpoints in tumors.13,14 In
many tumors, upregulated programmed death-ligand 1
PD-L1 expression can either be constitutive or induced
to evade immune surveillance. PD-1 expressed on activated
T cells can bind to its ligand PD-L1 on tumor cells,
leads to T cell exhaustion and downregulated immune
defense against tumors.10 By blocking or counteracting
the tumor mediated inhibition of T cell receptor activated
IL 2 production and T cell proliferation, ICIs can
potentially suppress the events that otherwise downregulate
a cellular immune response. This counteraction results
in a successful anti tumor T cell mediated immune
activity and antibodies raised against such PD-1 and PDL1
inhibitory axis can unleash activated tumor-reactive
T cells to promote durable anti-tumor responses in many
tumors. Thus, this biological rationale encouraged the
synergistic association of CTLA-4 inhibition, which facilitates
active immune response at the level of T-cell proliferation,
with PD-1 suppression, which modulates the
immune response at the level of the tumor micro-environment.
Since the disruption of PD-1–PD-L1 signaling
mediated by nivolumab can lead to restored antitumor
immunity, PD-L1 expression is associated with improved
overall survival in response to nivolumab therapy.11 This
anti-PD-1 antibody nivolumab, selectively blocks the interaction
of PD-1 (expressed on activated T cells) with its
ligands PD-L1 and PD-L2 (expressed on immune cells
and tumor cells) and thus counteracting the cellular immune
response pathways.12 Such discoveries led to the
approval of anti-PD1 antibodies (for example: pembrolizumab
and nivolumab) and anti-PD-L1 antibodies
(for example: atezolimumab) for the treatment of advanced
melanoma, NSCLC, RCC, head and neck squamous
52 Kidney Cancer Journal
carcinoma, Hodgkin’s lymphoma, and bladder cancer.13
Monotherapy or Combinatorial Therapy
of Immune Checkpoint Inhibitors
The ICI field is evolving rapidly with many clinical trials
already completed studying several checkpoint inhibitors
alone, in combination, or with other targeted
therapies. Since the approval of the CTLA-4 antibody ipilimumab
in patients with melanoma in 2011, several
PD-1/PD-L1 inhibitors including nivolumab, pembrolizumab,
atezolizumab, durvalumab, and avelumab as
well as the CTLA-4 inhibitor ipilimumab were investigated
for their anti-tumor efficacy.23 Nivolumab, a fully
humanized IgG4 anti-PD-1 that was developed in the
form of a monoclonal antibody directed at PD-1, became
the first ICI approved by FDA in 2015 for the treatment
of refractory mRCC. In a randomized, open-label, phase
III study CheckMate-025 (NCT01668784), a total of 821
advanced ccRCC patients who had received previous
treatment with one or two regimens of antiangiogenic
therapy were randomly assigned either nivolumab or
everolimus.8 The primary end point was overall survival
and the secondary end points included the objective response
rate and safety. Results showed that the objective
response rate (ORR) was greater with nivolumab than with
everolimus (25% vs. 5%; p < 0.001) and median PFS was
better with nivolumab than with everolimus (4.6 months vs
4.4 months; p = 0.11).8 Results indicated that the
nivolumab arm had 25.0 months median overall survival
(95% CI, 21.8 to not estimable), longer as compared to
only 19.6 months (95% CI, 17.6 to 23.1) in the
everolimus arm. Nivolumab’s overall survival benefit
was evident across prespecified subgroups, including subgroups
defined per region, MSKCC prognostic score, and
number of previous regimens of antiangiogenic therapy.
Only 19% of the patients receiving nivolumab experienced
grade 3 or 4 treatment-related adverse events as
compared to 37% of the patients receiving everolimus, and
only 8% requiring treatment discontinuation because of
maintenance
Comparator Sunitinib Sunitinuib Sunitinib Sunitinib
Primary endpoint Co-primary endpoint of OS, Co-primary endpoint of Co-primary endpoint of OS and PFS and OS
PFS, and ORR in OS and PFS PFS in patients with PD-L1 ≥1%
intermediate risk
Number of 1,096 861 886 915
Patients recruited
Median OS, NR vs. 37.9 NR vs NR Not reported 24-mo: 63% vs. 83%
months
Median PFA, 9.7 vs 9.7 15.1 vs 11.1 13.8 vs 7.2 12-mo: 86% vs 83%
months
Overall response 41 vs 34 59.3 vs 35.7 51.4 vs 25.7 37% vs 33%
rate, %