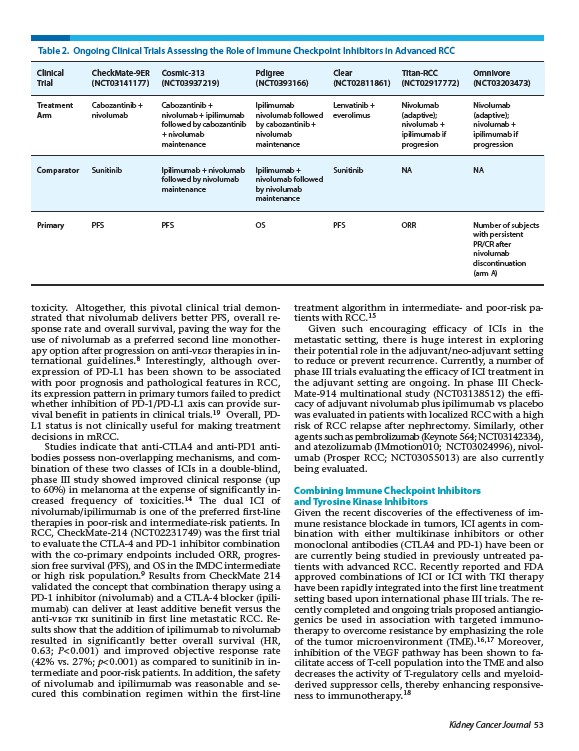
Table 2. Ongoing Clinical Trials Assessing the Role of Immune Checkpoint Inhibitors in Advanced RCC
Clinical CheckMate-9ER Cosmic-313 Pdigree Clear Titan-RCC Omnivore
Trial (NCT03141177) (NCT03937219) (NCT0393166) (NCT02811861) (NCT02917772) (NCT03203473)
Treatment Cabozantinib + Cabozantinib + Ipilimumab Lenvatinib + Nivolumab Nivolumab
Arm nivolumab nivolumab + ipilimumab nivolumab followed everolimus (adaptive); (adaptive);
followed by cabozantinib by cabozantinib + nivolumab + nivolumab +
+ nivolumab nivolumab ipilimumab if ipilimumab if
maintenance maintenance progresion progression
Comparator Sunitinib Ipilimumab + nivolumab Ipilimumab + Sunitinib NA NA
Kidney Cancer Journal 53
followed by nivolumab nivolumab followed
maintenance by nivolumab
toxicity. Altogether, this pivotal clinical trial demonstrated
that nivolumab delivers better PFS, overall response
rate and overall survival, paving the way for the
use of nivolumab as a preferred second line monotherapy
option after progression on anti-VEGF therapies in international
guidelines.8 Interestingly, although over-
expression of PD-L1 has been shown to be associated
with poor prognosis and pathological features in RCC,
its expression pattern in primary tumors failed to predict
whether inhibition of PD-1/PD-L1 axis can provide survival
benefit in patients in clinical trials.19 Overall, PDL1
status is not clinically useful for making treatment
decisions in mRCC.
Studies indicate that anti-CTLA4 and anti-PD1 antibodies
possess non-overlapping mechanisms, and combination
of these two classes of ICIs in a double-blind,
phase III study showed improved clinical response (up
to 60%) in melanoma at the expense of significantly increased
frequency of toxicities.14 The dual ICI of
nivolumab/ipilimumab is one of the preferred first-line
therapies in poor-risk and intermediate-risk patients. In
RCC, CheckMate-214 (NCT02231749) was the first trial
to evaluate the CTLA-4 and PD-1 inhibitor combination
with the co-primary endpoints included ORR, progression
free survival (PFS), and OS in the IMDC intermediate
or high risk population.9 Results from CheckMate 214
validated the concept that combination therapy using a
PD-1 inhibitor (nivolumab) and a CTLA-4 blocker (ipilimumab)
can deliver at least additive benefit versus the
anti-VEGF TKI sunitinib in first line metastatic RCC. Results
show that the addition of ipilimumab to nivolumab
resulted in significantly better overall survival (HR,
0.63; P < 0.001) and improved objective response rate
(42% vs. 27%; p < 0.001) as compared to sunitinib in intermediate
and poor-risk patients. In addition, the safety
of nivolumab and ipilimumab was reasonable and secured
this combination regimen within the first-line
treatment algorithm in intermediate- and poor-risk patients
with RCC.15
Given such encouraging efficacy of ICIs in the
metastatic setting, there is huge interest in exploring
their potential role in the adjuvant/neo-adjuvant setting
to reduce or prevent recurrence. Currently, a number of
phase III trials evaluating the efficacy of ICI treatment in
the adjuvant setting are ongoing. In phase III Check-
Mate-914 multinational study (NCT03138512) the efficacy
of adjuvant nivolumab plus ipilimumab vs placebo
was evaluated in patients with localized RCC with a high
risk of RCC relapse after nephrectomy. Similarly, other
agents such as pembrolizumab (Keynote 564; NCT03142334),
and atezolizumab (IMmotion010; NCT03024996), nivolumab
(Prosper RCC; NCT03055013) are also currently
being evaluated.
Combining Immune Checkpoint Inhibitors
and Tyrosine Kinase Inhibitors
Given the recent discoveries of the effectiveness of immune
resistance blockade in tumors, ICI agents in combination
with either multikinase inhibitors or other
monoclonal antibodies (CTLA4 and PD-1) have been or
are currently being studied in previously untreated patients
with advanced RCC. Recently reported and FDA
approved combinations of ICI or ICI with TKI therapy
have been rapidly integrated into the first line treatment
setting based upon international phase III trials. The recently
completed and ongoing trials proposed antiangiogenics
be used in association with targeted immuno-
therapy to overcome resistance by emphasizing the role
of the tumor microenvironment (TME).16,17 Moreover,
inhibition of the VEGF pathway has been shown to facilitate
access of T-cell population into the TME and also
decreases the activity of T-regulatory cells and myeloidderived
suppressor cells, thereby enhancing responsiveness
to immunotherapy.18
maintenance
Primary PFS PFS OS PFS ORR Number of subjects
with persistent
PR/CR after
nivolumab
discontinuation
(arm A)