Kidney Cancer Journal 59
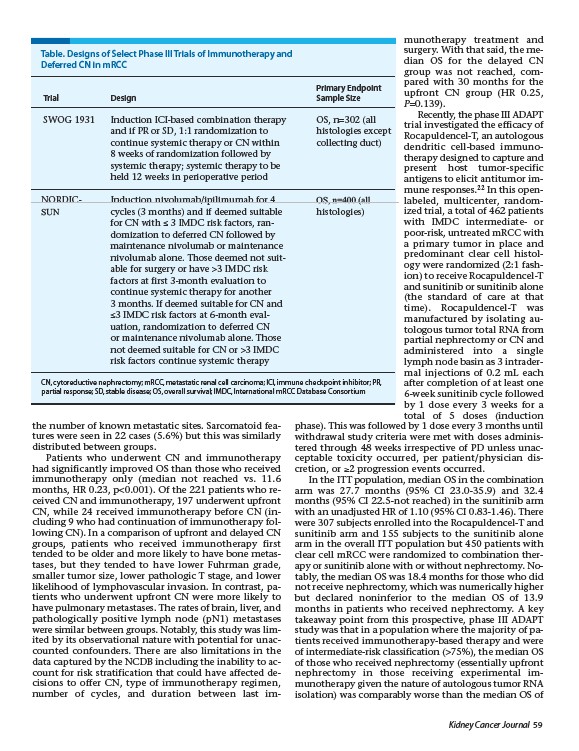
Table. Designs of Select Phase III Trials of Immunotherapy and
Deferred CN in mRCC
Primary Endpoint
Trial Design Sample Size
SWOG 1931 Induction ICI-based combination therapy OS, n=302 (all
and if PR or SD, 1:1 randomization to histologies except
continue systemic therapy or CN within collecting duct)
8 weeks of randomization followed by
systemic therapy; systemic therapy to be
held 12 weeks in perioperative period
NORDIC- Induction nivolumab/ipilimumab for 4 OS, n=400 (all
SUN cycles (3 months) and if deemed suitable histologies)
the number of known metastatic sites. Sarcomatoid features
were seen in 22 cases (5.6%) but this was similarly
distributed between groups.
Patients who underwent CN and immunotherapy
had significantly improved OS than those who received
immunotherapy only (median not reached vs. 11.6
months, HR 0.23, p<0.001). Of the 221 patients who received
CN and immunotherapy, 197 underwent upfront
CN, while 24 received immunotherapy before CN (including
9 who had continuation of immunotherapy following
CN). In a comparison of upfront and delayed CN
groups, patients who received immunotherapy first
tended to be older and more likely to have bone metastases,
but they tended to have lower Fuhrman grade,
smaller tumor size, lower pathologic T stage, and lower
likelihood of lymphovascular invasion. In contrast, patients
who underwent upfront CN were more likely to
have pulmonary metastases. The rates of brain, liver, and
pathologically positive lymph node (pN1) metastases
were similar between groups. Notably, this study was limited
by its observational nature with potential for unaccounted
confounders. There are also limitations in the
data captured by the NCDB including the inability to account
for risk stratification that could have affected decisions
to offer CN, type of immunotherapy regimen,
number of cycles, and duration between last immunotherapy
treatment and
surgery. With that said, the median
OS for the delayed CN
group was not reached, compared
with 30 months for the
upfront CN group (HR 0.25,
P=0.139).
Recently, the phase III ADAPT
trial investigated the efficacy of
Rocapuldencel-T, an autologous
dendritic cell-based immunotherapy
designed to capture and
present host tumor-specific
antigens to elicit antitumor immune
responses.22 In this openlabeled,
multicenter, random-
ized trial, a total of 462 patients
with IMDC intermediate- or
poor-risk, untreated mRCC with
a primary tumor in place and
predominant clear cell histology
were randomized (2:1 fashion)
to receive Rocapuldencel-T
and sunitinib or sunitinib alone
(the standard of care at that
time). Rocapuldencel-T was
manufactured by isolating autologous
tumor total RNA from
partial nephrectomy or CN and
administered into a single
lymph node basin as 3 intradermal
injections of 0.2 mL each
after completion of at least one
6-week sunitinib cycle followed
by 1 dose every 3 weeks for a
total of 5 doses (induction
phase). This was followed by 1 dose every 3 months until
withdrawal study criteria were met with doses administered
through 48 weeks irrespective of PD unless unacceptable
toxicity occurred, per patient/physician dis-
cretion, or ��2 progression events occurred.
In the ITT population, median OS in the combination
arm was 27.7 months (95% CI 23.0-35.9) and 32.4
months (95% CI 22.5-not reached) in the sunitinib arm
with an unadjusted HR of 1.10 (95% CI 0.83-1.46). There
were 307 subjects enrolled into the Rocapuldencel-T and
sunitinib arm and 155 subjects to the sunitinib alone
arm in the overall ITT population but 450 patients with
clear cell mRCC were randomized to combination therapy
or sunitinib alone with or without nephrectomy. Notably,
the median OS was 18.4 months for those who did
not receive nephrectomy, which was numerically higher
but declared noninferior to the median OS of 13.9
months in patients who received nephrectomy. A key
takeaway point from this prospective, phase III ADAPT
study was that in a population where the majority of patients
received immunotherapy-based therapy and were
of intermediate-risk classification (>75%), the median OS
of those who received nephrectomy (essentially upfront
nephrectomy in those receiving experimental immunotherapy
given the nature of autologous tumor RNA
isolation) was comparably worse than the median OS of
for CN with �� 3 IMDC risk factors, ran-
domization to deferred CN followed by
maintenance nivolumab or maintenance
nivolumab alone. Those deemed not suit-
able for surgery or have >3 IMDC risk
factors at first 3-month evaluation to
continue systemic therapy for another
3 months. If deemed suitable for CN and
��3 IMDC risk factors at 6-month eval-
uation, randomization to deferred CN
or maintenance nivolumab alone. Those
not deemed suitable for CN or >3 IMDC
risk factors continue systemic therapy
CN, cytoreductive nephrectomy; mRCC, metastatic renal cell carcinoma; ICI, immune checkpoint inhibitor; PR,
partial response; SD, stable disease; OS, overall survival; IMDC, International mRCC Database Consortium