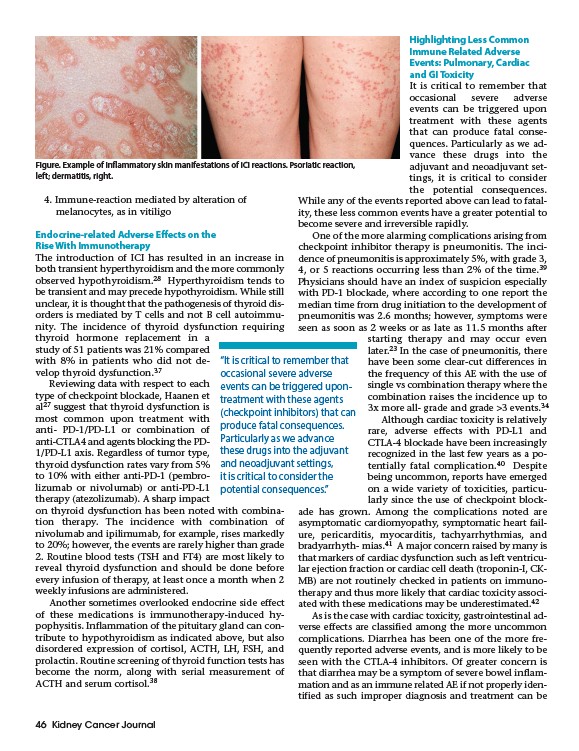
Figure. Example of inflammatory skin manifestations of ICI reactions. Psoriatic reaction,
left; dermatitis, right.
4. Immune-reaction mediated by alteration of
melanocytes, as in vitiligo
Endocrine-related Adverse Effects on the
Rise With Immunotherapy
The introduction of ICI has resulted in an increase in
both transient hyperthyroidism and the more commonly
observed hypothyroidism.28 Hyperthyroidism tends to
be transient and may precede hypothyroidism. While still
unclear, it is thought that the pathogenesis of thyroid disorders
is mediated by T cells and not B cell autoimmunity.
The incidence of thyroid dysfunction requiring
thyroid hormone replacement in a
study of 51 patients was 21% compared
with 8% in patients who did not develop
thyroid dysfunction.37
Reviewing data with respect to each
type of checkpoint blockade, Haanen et
al27 suggest that thyroid dysfunction is
most common upon treatment with
anti- PD-1/PD-L1 or combination of
anti-CTLA4 and agents blocking the PD-
1/PD-L1 axis. Regardless of tumor type,
thyroid dysfunction rates vary from 5%
to 10% with either anti-PD-1 (pembrolizumab
or nivolumab) or anti-PD-L1
“It is critical to remember that
occasional severe adverse
events can be triggered upontreatment
with these agents
(checkpoint inhibitors) that can
produce fatal consequences.
Particularly as we advance
these drugs into the adjuvant
and neoadjuvant settings,
it is critical to consider the
potential consequences.”
therapy (atezolizumab). A sharp impact
on thyroid dysfunction has been noted with combination
therapy. The incidence with combination of
nivolumab and ipilimumab, for example, rises markedly
to 20%; however, the events are rarely higher than grade
2. Routine blood tests (TSH and FT4) are most likely to
reveal thyroid dysfunction and should be done before
every infusion of therapy, at least once a month when 2
weekly infusions are administered.
Another sometimes overlooked endocrine side effect
of these medications is immunotherapy-induced hypophysitis.
Inflammation of the pituitary gland can contribute
to hypothyroidism as indicated above, but also
disordered expression of cortisol, ACTH, LH, FSH, and
prolactin. Routine screening of thyroid function tests has
become the norm, along with serial measurement of
ACTH and serum cortisol.38
46 Kidney Cancer Journal
Highlighting Less Common
Immune Related Adverse
Events: Pulmonary, Cardiac
and GI Toxicity
It is critical to remember that
occasional severe adverse
events can be triggered upon
treatment with these agents
that can produce fatal consequences.
Particularly as we advance
these drugs into the
adjuvant and neoadjuvant settings,
it is critical to consider
the potential consequences.
While any of the events reported above can lead to fatality,
these less common events have a greater potential to
become severe and irreversible rapidly.
One of the more alarming complications arising from
checkpoint inhibitor therapy is pneumonitis. The incidence
of pneumonitis is approximately 5%, with grade 3,
4, or 5 reactions occurring less than 2% of the time.39
Physicians should have an index of suspicion especially
with PD-1 blockade, where according to one report the
median time from drug initiation to the development of
pneumonitis was 2.6 months; however, symptoms were
seen as soon as 2 weeks or as late as 11.5 months after
starting therapy and may occur even
later.23 In the case of pneumonitis, there
have been some clear-cut differences in
the frequency of this AE with the use of
single vs combination therapy where the
combination raises the incidence up to
3x more all- grade and grade >3 events.34
Although cardiac toxicity is relatively
rare, adverse effects with PD-L1 and
CTLA-4 blockade have been increasingly
recognized in the last few years as a potentially
fatal complication.40 Despite
being uncommon, reports have emerged
on a wide variety of toxicities, particularly
since the use of checkpoint blockade
has grown. Among the complications noted are
asymptomatic cardiomyopathy, symptomatic heart failure,
pericarditis, myocarditis, tachyarrhythmias, and
bradyarrhyth- mias.41 A major concern raised by many is
that markers of cardiac dysfunction such as left ventricular
ejection fraction or cardiac cell death (troponin-I, CKMB)
are not routinely checked in patients on immunotherapy
and thus more likely that cardiac toxicity associated
with these medications may be underestimated.42
As is the case with cardiac toxicity, gastrointestinal adverse
effects are classified among the more uncommon
complications. Diarrhea has been one of the more frequently
reported adverse events, and is more likely to be
seen with the CTLA-4 inhibitors. Of greater concern is
that diarrhea may be a symptom of severe bowel inflammation
and as an immune related AE if not properly identified
as such improper diagnosis and treatment can be