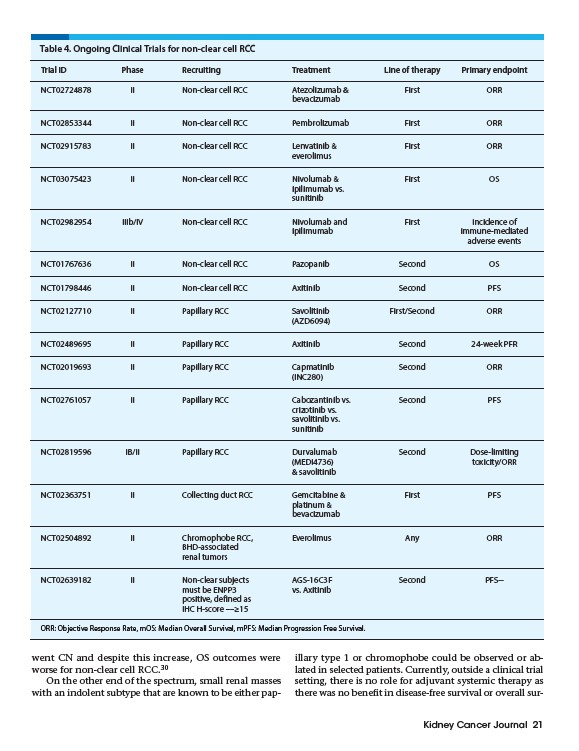
Table 4. Ongoing Clinical Trials for non-clear cell RCC
Trial ID Phase Recruiting Treatment Line of therapy Primary endpoint
NCT02724878 II Non-clear cell RCC Atezolizumab & First ORR
NCT02853344 II Non-clear cell RCC Pembrolizumab First ORR
NCT02915783 II Non-clear cell RCC Lenvatinib & First ORR
NCT03075423 II Non-clear cell RCC Nivolumab & First OS
NCT02982954 IIIb/IV Non-clear cell RCC Nivolumab and First Incidence of
ipilimumab immune-mediated
adverse events
NCT01767636 II Non-clear cell RCC Pazopanib Second OS
NCT01798446 II Non-clear cell RCC Axitinib Second PFS
NCT02127710 II Papillary RCC Savolitinib First/Second ORR
NCT02489695 II Papillary RCC Axitinib Second 24-week PFR
NCT02019693 II Papillary RCC Capmatinib Second ORR
NCT02761057 II Papillary RCC Cabozantinib vs. Second PFS
NCT02819596 IB/II Papillary RCC Durvalumab Second Dose-limiting
(MEDI4736) toxicity/ORR
& savolitinib
NCT02363751 II Collecting duct RCC Gemcitabine & First PFS
NCT02504892 II Chromophobe RCC, Everolimus Any ORR
Kidney Cancer Journal 21
went CN and despite this increase, OS outcomes were
worse for non-clear cell RCC.30
On the other end of the spectrum, small renal masses
with an indolent subtype that are known to be either papillary
bevacizumab
everolimus
ipilimumab vs.
sunitinib
(AZD6094)
(INC280)
crizotinib vs.
savolitinib vs.
sunitinib
platinum &
bevacizumab
type 1 or chromophobe could be observed or ablated
in selected patients. Currently, outside a clinical trial
setting, there is no role for adjuvant systemic therapy as
there was no benefit in disease-free survival or overall sur-
BHD-associated
renal tumors
NCT02639182 II Non-clear subjects AGS-16C3F Second PFS - -
must be ENPP3 vs. Axitinib
positive, defined as
IHC H-score ––≥15
ORR: Objective Response Rate, mOS: Median Overall Survival, mPFS: Median Progression Free Survival.