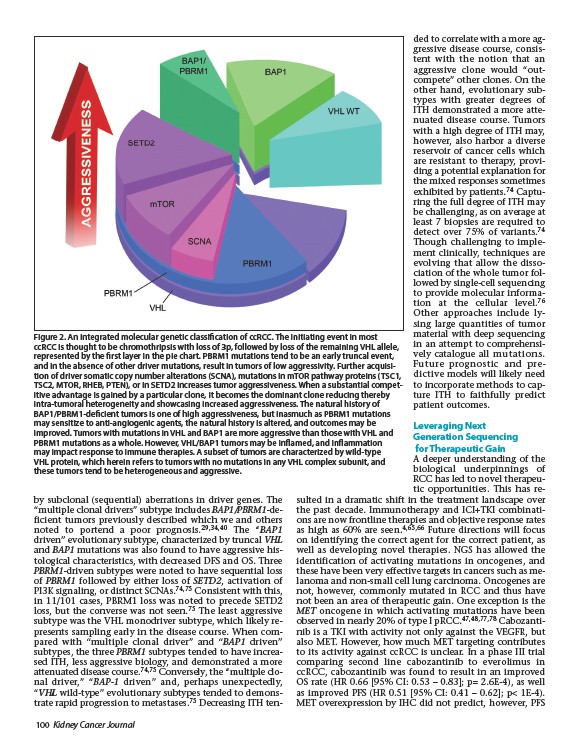
Figure 2. An integrated molecular genetic classification of ccRCC. The initiating event in most
ccRCC is thought to be chromothripsis with loss of 3p, followed by loss of the remaining VHL allele,
represented by the first layer in the pie chart. PBRM1 mutations tend to be an early truncal event,
and in the absence of other driver mutations, result in tumors of low aggressivity. Further acquisition
of driver somatic copy number alterations (SCNA), mutations in mTOR pathway proteins (TSC1,
TSC2, MTOR, RHEB, PTEN), or in SETD2 increases tumor aggressiveness. When a substantial competitive
advantage is gained by a particular clone, it becomes the dominant clone reducing thereby
intra-tumoral heterogeneity and showcasing increased aggressiveness. The natural history of
BAP1/PBRM1-deficient tumors is one of high aggressiveness, but inasmuch as PBRM1 mutations
may sensitize to anti-angiogenic agents, the natural history is altered, and outcomes may be
improved. Tumors with mutations in VHL and BAP1 are more aggressive than those with VHL and
PBRM1 mutations as a whole. However, VHL/BAP1 tumors may be inflamed, and inflammation
may impact response to immune therapies. A subset of tumors are characterized by wild-type
VHL protein, which herein refers to tumors with no mutations in any VHL complex subunit, and
these tumors tend to be heterogeneous and aggressive.
by subclonal (sequential) aberrations in driver genes. The
“multiple clonal drivers” subtype includes BAP1/PBRM1-deficient
tumors previously described which we and others
noted to portend a poor prognosis.29,34,40 The “BAP1
driven” evolutionary subtype, characterized by truncal VHL
and BAP1 mutations was also found to have aggressive histological
characteristics, with decreased DFS and OS. Three
PBRM1-driven subtypes were noted to have sequential loss
of PBRM1 followed by either loss of SETD2, activation of
PI3K signaling, or distinct SCNAs.74,75 Consistent with this,
in 11/101 cases, PBRM1 loss was noted to precede SETD2
loss, but the converse was not seen.75 The least aggressive
subtype was the VHL monodriver subtype, which likely represents
sampling early in the disease course. When compared
with “multiple clonal driver” and “BAP1 driven”
subtypes, the three PBRM1 subtypes tended to have increased
ITH, less aggressive biology, and demonstrated a more
attenuated disease course.74,75 Conversely, the “multiple clonal
driver,” “BAP-1 driven” and, perhaps unexpectedly,
“VHL wild-type” evolutionary subtypes tended to demonstrate
rapid progression to metastases.75 Decreasing ITH tended
100 Kidney Cancer Journal
to correlate with a more aggressive
disease course, consistent
with the notion that an
aggressive clone would “outcompete”
other clones. On the
other hand, evolutionary subtypes
with greater degrees of
ITH demonstrated a more attenuated
disease course. Tumors
with a high degree of ITH may,
however, also harbor a diverse
reservoir of cancer cells which
are resistant to therapy, providing
a potential explanation for
the mixed responses sometimes
exhibited by patients.74 Capturing
the full degree of ITH may
be challenging, as on average at
least 7 biopsies are required to
detect over 75% of variants.74
Though challenging to implement
clinically, techniques are
evolving that allow the dissociation
of the whole tumor followed
by single-cell sequencing
to provide molecular information
at the cellular level.76
Other approaches include lysing
large quantities of tumor
material with deep sequencing
in an attempt to comprehensively
catalogue all mutations.
Future prognostic and predictive
models will likely need
to incorporate methods to capture
ITH to faithfully predict
patient outcomes.
Leveraging Next
Generation Sequencing
for Therapeutic Gain
A deeper understanding of the
biological underpinnings of
RCC has led to novel therapeutic
opportunities. This has resulted
in a dramatic shift in the treatment landscape over
the past decade. Immunotherapy and ICI+TKI combinations
are now frontline therapies and objective response rates
as high as 60% are seen.4,65,66 Future directions will focus
on identifying the correct agent for the correct patient, as
well as developing novel therapies. NGS has allowed the
identification of activating mutations in oncogenes, and
these have been very effective targets in cancers such as melanoma
and non-small cell lung carcinoma. Oncogenes are
not, however, commonly mutated in RCC and thus have
not been an area of therapeutic gain. One exception is the
MET oncogene in which activating mutations have been
observed in nearly 20% of type I pRCC.47,48,77,78 Cabozantinib
is a TKI with activity not only against the VEGFR, but
also MET. However, how much MET targeting contributes
to its activity against ccRCC is unclear. In a phase III trial
comparing second line cabozantinib to everolimus in
ccRCC, cabozantinib was found to result in an improved
OS rate (HR 0.66 95% CI: 0.53 – 0.83; p= 2.6E-4), as well
as improved PFS (HR 0.51 95% CI: 0.41 – 0.62; p< 1E-4).
MET overexpression by IHC did not predict, however, PFS