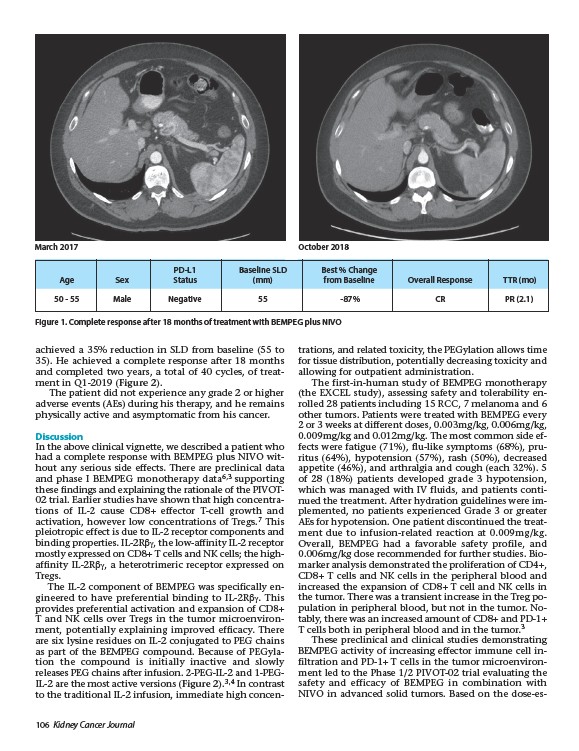
March 2017 October 2018
PD-L1 Baseline SLD Best % Change
Age Sex Status (mm) from Baseline Overall Response TTR (mo)
Figure 1. Complete response after 18 months of treatment with BEMPEG plus NIVO
achieved a 35% reduction in SLD from baseline (55 to
35). He achieved a complete response after 18 months
and completed two years, a total of 40 cycles, of treatment
in Q1-2019 (Figure 2).
The patient did not experience any grade 2 or higher
adverse events (AEs) during his therapy, and he remains
physically active and asymptomatic from his cancer.
Discussion
In the above clinical vignette, we described a patient who
had a complete response with BEMPEG plus NIVO without
any serious side effects. There are preclinical data
and phase I BEMPEG monotherapy data6,3 supporting
these findings and explaining the rationale of the PIVOT-
02 trial. Earlier studies have shown that high concentrations
of IL-2 cause CD8+ effector T-cell growth and
activation, however low concentrations of Tregs.7 This
pleiotropic effect is due to IL-2 receptor components and
binding properties. IL-2Rbg, the low-affinity IL-2 receptor
mostly expressed on CD8+ T cells and NK cells; the highaffinity
IL-2Rbg, a heterotrimeric receptor expressed on
Tregs.
The IL-2 component of BEMPEG was specifically engineered
to have preferential binding to IL-2Rbg. This
provides preferential activation and expansion of CD8+
T and NK cells over Tregs in the tumor microenvironment,
potentially explaining improved efficacy. There
are six lysine residues on IL-2 conjugated to PEG chains
as part of the BEMPEG compound. Because of PEGylation
the compound is initially inactive and slowly
releases PEG chains after infusion. 2-PEG-IL-2 and 1-PEGIL
2 are the most active versions (Figure 2).3,4 In contrast
to the traditional IL-2 infusion, immediate high concentrations,
106 Kidney Cancer Journal
and related toxicity, the PEGylation allows time
for tissue distribution, potentially decreasing toxicity and
allowing for outpatient administration.
The first-in-human study of BEMPEG monotherapy
(the EXCEL study), assessing safety and tolerability enrolled
28 patients including 15 RCC, 7 melanoma and 6
other tumors. Patients were treated with BEMPEG every
2 or 3 weeks at different doses, 0.003mg/kg, 0.006mg/kg,
0.009mg/kg and 0.012mg/kg. The most common side effects
were fatigue (71%), flu-like symptoms (68%), pruritus
(64%), hypotension (57%), rash (50%), decreased
appetite (46%), and arthralgia and cough (each 32%). 5
of 28 (18%) patients developed grade 3 hypotension,
which was managed with IV fluids, and patients continued
the treatment. After hydration guidelines were implemented,
no patients experienced Grade 3 or greater
AEs for hypotension. One patient discontinued the treatment
due to infusion-related reaction at 0.009mg/kg.
Overall, BEMPEG had a favorable safety profile, and
0.006mg/kg dose recommended for further studies. Biomarker
analysis demonstrated the proliferation of CD4+,
CD8+ T cells and NK cells in the peripheral blood and
increased the expansion of CD8+ T cell and NK cells in
the tumor. There was a transient increase in the Treg population
in peripheral blood, but not in the tumor. Notably,
there was an increased amount of CD8+ and PD-1+
T cells both in peripheral blood and in the tumor.3
These preclinical and clinical studies demonstrating
BEMPEG activity of increasing effector immune cell infiltration
and PD-1+ T cells in the tumor microenvironment
led to the Phase 1/2 PIVOT-02 trial evaluating the
safety and efficacy of BEMPEG in combination with
NIVO in advanced solid tumors. Based on the dose-es-
50 - 55 Male Negative 55 -87% CR PR (2.1)