Kidney Cancer Journal 107
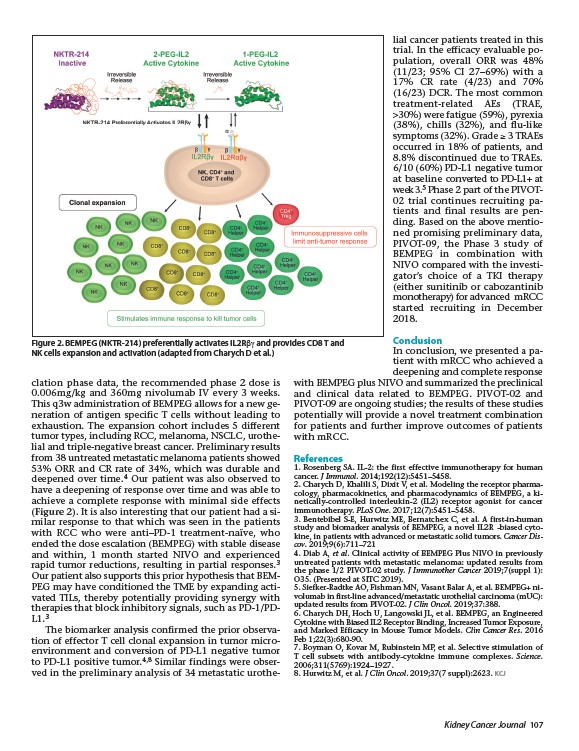
Figure 2. BEMPEG (NKTR-214) preferentially activates IL2Rbg and provides CD8 T and
NK cells expansion and activation (adapted from Charych D et al.)
clation phase data, the recommended phase 2 dose is
0.006mg/kg and 360mg nivolumab IV every 3 weeks.
This q3w administration of BEMPEG allows for a new generation
of antigen specific T cells without leading to
exhaustion. The expansion cohort includes 5 different
tumor types, including RCC, melanoma, NSCLC, urothelial
and triple-negative breast cancer. Preliminary results
from 38 untreated metastatic melanoma patients showed
53% ORR and CR rate of 34%, which was durable and
deepened over time.4 Our patient was also observed to
have a deepening of response over time and was able to
achieve a complete response with minimal side effects
(Figure 2). It is also interesting that our patient had a similar
response to that which was seen in the patients
with RCC who were anti–PD-1 treatment-naïve, who
ended the dose escalation (BEMPEG) with stable disease
and within, 1 month started NIVO and experienced
rapid tumor reductions, resulting in partial responses.3
Our patient also supports this prior hypothesis that BEMPEG
may have conditioned the TME by expanding activated
TILs, thereby potentially providing synergy with
therapies that block inhibitory signals, such as PD-1/PDL1.3
The biomarker analysis confirmed the prior observation
of effector T cell clonal expansion in tumor microenvironment
and conversion of PD-L1 negative tumor
to PD-L1 positive tumor.4,8 Similar findings were observed
in the preliminary analysis of 34 metastatic urothelial
cancer patients treated in this
trial. In the efficacy evaluable population,
overall ORR was 48%
(11/23; 95% CI 27–69%) with a
17% CR rate (4/23) and 70%
(16/23) DCR. The most common
treatment-related AEs (TRAE,
>30%) were fatigue (59%), pyrexia
(38%), chills (32%), and flu-like
symptoms (32%). Grade �� 3 TRAEs
occurred in 18% of patients, and
8.8% discontinued due to TRAEs.
6/10 (60%) PD-L1 negative tumor
at baseline converted to PD-L1+ at
week 3.5 Phase 2 part of the PIVOT-
02 trial continues recruiting patients
and final results are pen-
ding. Based on the above mentioned
promising preliminary data,
PIVOT-09, the Phase 3 study of
BEMPEG in combination with
NIVO compared with the investigator’s
choice of a TKI therapy
(either sunitinib or cabozantinib
monotherapy) for advanced mRCC
started recruiting in December
2018.
Conclusion
In conclusion, we presented a patient
with mRCC who achieved a
deepening and complete response
with BEMPEG plus NIVO and summarized the preclinical
and clinical data related to BEMPEG. PIVOT-02 and
PIVOT-09 are ongoing studies; the results of these studies
potentially will provide a novel treatment combination
for patients and further improve outcomes of patients
with mRCC.
References
1. Rosenberg SA. IL-2: the first effective immunotherapy for human
cancer. J Immunol. 2014;192(12):5451–5458.
2. Charych D, Khalili S, Dixit V, et al. Modeling the receptor pharmacology,
pharmacokinetics, and pharmacodynamics of BEMPEG, a kinetically
controlled interleukin-2 (IL2) receptor agonist for cancer
immunotherapy. PLoS One. 2017;12(7):5451–5458.
3. Bentebibel S-E, Hurwitz ME, Bernatchex C, et al. A first-in-human
study and biomarker analysis of BEMPEG, a novel IL2R -biased cytokine,
in patients with advanced or metastatic solid tumors. Cancer Discov.
2019;9(6):711–721
4. Diab A, et al. Clinical activity of BEMPEG Plus NIVO in previously
untreated patients with metastatic melanoma: updated results from
the phase 1/2 PIVOT-02 study. J Immunother Cancer 2019;7(suppl 1):
O35. (Presented at SITC 2019).
5. Siefker-Radtke AO, Fishman MN, Vasant Balar A, et al. BEMPEG+ nivolumab
in first-line advanced/metastatic urothelial carcinoma (mUC):
updated results from PIVOT-02. J Clin Oncol. 2019;37:388.
6. Charych DH, Hoch U, Langowski JL, et al. BEMPEG, an Engineered
Cytokine with Biased IL2 Receptor Binding, Increased Tumor Exposure,
and Marked Efficacy in Mouse Tumor Models. Clin Cancer Res. 2016
Feb 1;22(3):680-90.
7. Boyman O, Kovar M, Rubinstein MP, et al. Selective stimulation of
T cell subsets with antibody-cytokine immune complexes. Science.
2006;311(5769):1924–1927.
8. Hurwitz M, et al. J Clin Oncol. 2019;37(7 suppl):2623. KCJ