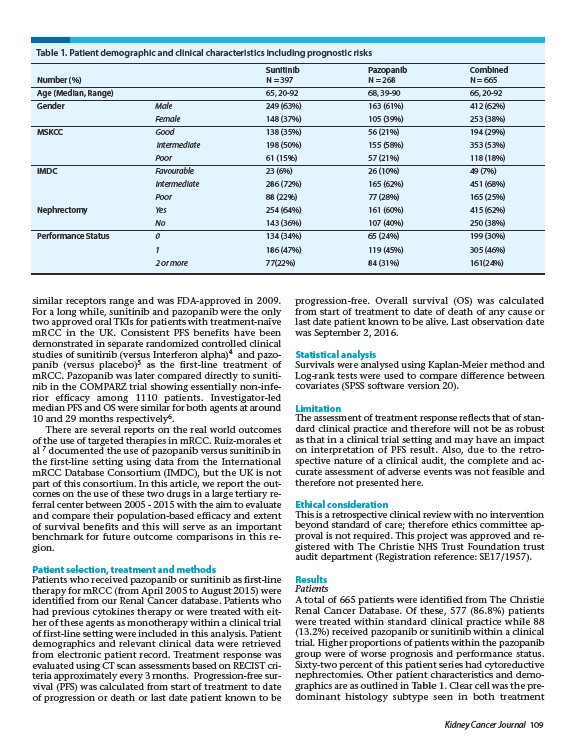
Table 1. Patient demographic and clinical characteristics including prognostic risks
Sunitinib Pazopanib Combined
Number (%) N = 397 N = 268 N = 665
Age (Median, Range) 65, 20-92 68, 39-90 66, 20-92
Gender Male 249 (63%) 163 (61%) 412 (62%)
Female 148 (37%) 105 (39%) 253 (38%)
MSKCC Good 138 (35%) 56 (21%) 194 (29%)
Intermediate 198 (50%) 155 (58%) 353 (53%)
Poor 61 (15%) 57 (21%) 118 (18%)
IMDC Favourable 23 (6%) 26 (10%) 49 (7%)
Intermediate 286 (72%) 165 (62%) 451 (68%)
Poor 88 (22%) 77 (28%) 165 (25%)
Nephrectomy Yes 254 (64%) 161 (60%) 415 (62%)
No 143 (36%) 107 (40%) 250 (38%)
Performance Status 0 134 (34%) 65 (24%) 199 (30%)
1 186 (47%) 119 (45%) 305 (46%)
2 or more 77(22%) 84 (31%) 161(24%)
similar receptors range and was FDA-approved in 2009.
For a long while, sunitinib and pazopanib were the only
two approved oral TKIs for patients with treatment-naïve
mRCC in the UK. Consistent PFS benefits have been
demonstrated in separate randomized controlled clinical
studies of sunitinib (versus Interferon alpha)4 and pazopanib
Kidney Cancer Journal 109
(versus placebo)5 as the first-line treatment of
mRCC. Pazopanib was later compared directly to sunitinib
in the COMPARZ trial showing essentially non-inferior
efficacy among 1110 patients. Investigator-led
median PFS and OS were similar for both agents at around
10 and 29 months respectively6.
There are several reports on the real world outcomes
of the use of targeted therapies in mRCC. Ruiz-morales et
al 7 documented the use of pazopanib versus sunitinib in
the first-line setting using data from the International
mRCC Database Consortium (IMDC), but the UK is not
part of this consortium. In this article, we report the outcomes
on the use of these two drugs in a large tertiary referral
center between 2005 - 2015 with the aim to evaluate
and compare their population-based efficacy and extent
of survival benefits and this will serve as an important
benchmark for future outcome comparisons in this region.
Patient selection, treatment and methods
Patients who received pazopanib or sunitinib as first-line
therapy for mRCC (from April 2005 to August 2015) were
identified from our Renal Cancer database. Patients who
had previous cytokines therapy or were treated with either
of these agents as monotherapy within a clinical trial
of first-line setting were included in this analysis. Patient
demographics and relevant clinical data were retrieved
from electronic patient record. Treatment response was
evaluated using CT scan assessments based on RECIST criteria
approximately every 3 months. Progression-free survival
(PFS) was calculated from start of treatment to date
of progression or death or last date patient known to be
progression-free. Overall survival (OS) was calculated
from start of treatment to date of death of any cause or
last date patient known to be alive. Last observation date
was September 2,2016.
Statistical analysis
Survivals were analysed using Kaplan-Meier method and
Log-rank tests were used to compare difference between
covariates (SPSS software version 20).
Limitation
The assessment of treatment response reflects that of standard
clinical practice and therefore will not be as robust
as that in a clinical trial setting and may have an impact
on interpretation of PFS result. Also, due to the retrospective
nature of a clinical audit, the complete and accurate
assessment of adverse events was not feasible and
therefore not presented here.
Ethical consideration
This is a retrospective clinical review with no intervention
beyond standard of care; therefore ethics committee approval
is not required. This project was approved and registered
with The Christie NHS Trust Foundation trust
audit department (Registration reference: SE17/1957).
Results
Patients
A total of 665 patients were identified from The Christie
Renal Cancer Database. Of these, 577 (86.8%) patients
were treated within standard clinical practice while 88
(13.2%) received pazopanib or sunitinib within a clinical
trial. Higher proportions of patients within the pazopanib
group were of worse prognosis and performance status.
Sixty-two percent of this patient series had cytoreductive
nephrectomies. Other patient characteristics and demographics
are as outlined in Table 1. Clear cell was the predominant
histology subtype seen in both treatment