100
80
60
40
20
0
-20
-40
-60
-80
-100
130
120
110
100
90
80
70
60
50
40
30
20
10
0
-10
-20
-30
-40
-50
-60
-70
-80
-90
-100
-110
A
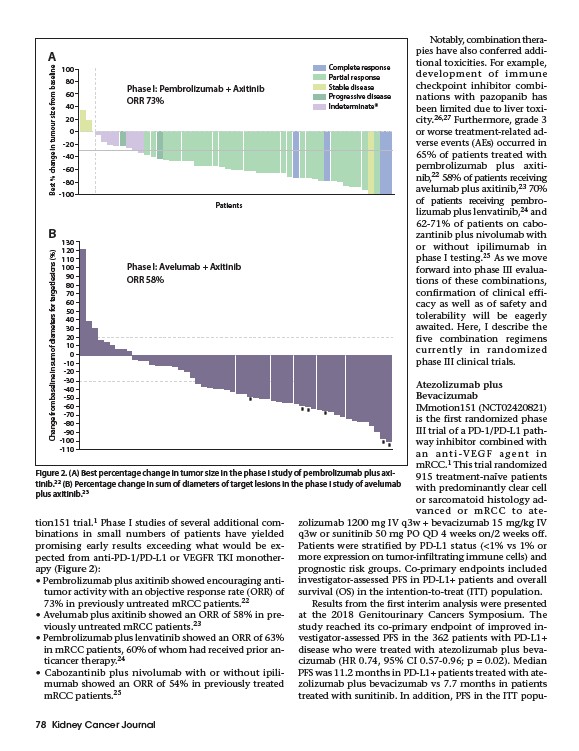
Figure 2. (A) Best percentage change in tumor size in the phase I study of pembrolizumab plus axitinib.
22 (B) Percentage change in sum of diameters of target lesions in the phase I study of avelumab
tion151 trial.1 Phase I studies of several additional combinations
in small numbers of patients have yielded
promising early results exceeding what would be expected
from anti-PD-1/PD-L1 or VEGFR TKI monotherapy
(Figure 2):
• Pembrolizumab plus axitinib showed encouraging antitumor
activity with an objective response rate (ORR) of
73% in previously untreated mRCC patients.22
• Avelumab plus axitinib showed an ORR of 58% in previously
untreated mRCC patients.23
• Pembrolizumab plus lenvatinib showed an ORR of 63%
in mRCC patients, 60% of whom had received prior anticancer
therapy.24
• Cabozantinib plus nivolumab with or without ipilimumab
showed an ORR of 54% in previously treated
mRCC patients.25
78 Kidney Cancer Journal
Notably, combination therapies
have also conferred additional
toxicities. For example,
development of immune
checkpoint inhibitor combinations
with pazopanib has
been limited due to liver toxicity.
26,27 Furthermore, grade 3
or worse treatment-related adverse
events (AEs) occurred in
65% of patients treated with
pembrolizumab plus axitinib,
22 58% of patients receiving
avelumab plus axitinib,23 70%
of patients receiving pembrolizumab
plus lenvatinib,24 and
62-71% of patients on cabozantinib
plus nivolumab with
or without ipilimumab in
phase I testing.25 As we move
forward into phase III evaluations
of these combinations,
confirmation of clinical efficacy
as well as of safety and
tolerability will be eagerly
awaited. Here, I describe the
five combination regimens
currently in randomized
phase III clinical trials.
Atezolizumab plus
Bevacizumab
IMmotion151 (NCT02420821)
is the first randomized phase
III trial of a PD-1/PD-L1 pathway
inhibitor combined with
an anti-VEGF agent in
mRCC.1 This trial randomized
915 treatment-naïve patients
with predominantly clear cell
or sarcomatoid histology advanced
or mRCC to atezolizumab
1200 mg IV q3w + bevacizumab 15 mg/kg IV
q3w or sunitinib 50 mg PO QD 4 weeks on/2 weeks off.
Patients were stratified by PD-L1 status (<1% vs 1% or
more expression on tumor-infiltrating immune cells) and
prognostic risk groups. Co-primary endpoints included
investigator-assessed PFS in PD-L1+ patients and overall
survival (OS) in the intention-to-treat (ITT) population.
Results from the first interim analysis were presented
at the 2018 Genitourinary Cancers Symposium. The
study reached its co-primary endpoint of improved investigator
assessed PFS in the 362 patients with PD-L1+
disease who were treated with atezolizumab plus bevacizumab
(HR 0.74, 95% CI 0.57-0.96; p = 0.02). Median
PFS was 11.2 months in PD-L1+ patients treated with atezolizumab
plus bevacizumab vs 7.7 months in patients
treated with sunitinib. In addition, PFS in the ITT popu-
plus axitinib.23
Patients
Change from baseline in sum of diameters for target lesions (%) Best % change in tumour size from baseline
B
Phase I: Pembrolizumab + Axitinib
ORR 73%
Phase I: Avelumab + Axitinib
ORR 58%
Complete response
Partial response
Stable disease
Progressive disease
Indeterminate*
*
* * *
* *