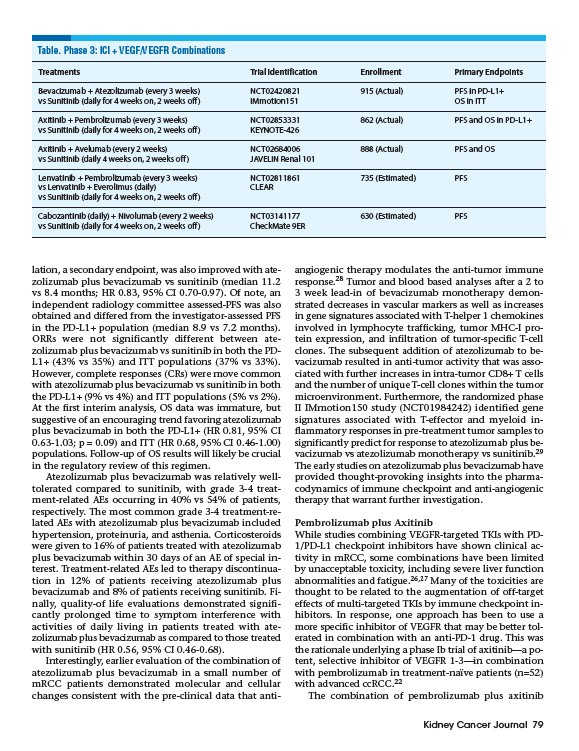
Table. Phase 3: ICI + VEGF/VEGFR Combinations
Treatments Trial Identification Enrollment Primary Endpoints
Bevacizumab + Atezolizumab (every 3 weeks) NCT02420821 915 (Actual) PFS in PD-L1+
vs Sunitinib (daily for 4 weeks on, 2 weeks off) IMmotion151 OS in ITT
Axitinib + Pembrolizumab (every 3 weeks) NCT02853331 862 (Actual) PFS and OS in PD-L1+
vs Sunitinib (daily for 4 weeks on, 2 weeks off) KEYNOTE-426
Axitinib + Avelumab (every 2 weeks) NCT02684006 888 (Actual) PFS and OS
vs Sunitinib (daily 4 weeks on, 2 weeks off) JAVELIN Renal 101
Lenvatinib + Pembrolizumab (every 3 weeks) NCT02811861 735 (Estimated) PFS
vs Lenvatinib + Everolimus (daily) CLEAR
vs Sunitinib (daily for 4 weeks on, 2 weeks off)
Cabozantinib (daily) + Nivolumab (every 2 weeks) NCT03141177 630 (Estimated) PFS
vs Sunitinib (daily for 4 weeks on, 2 weeks off) CheckMate 9ER
Kidney Cancer Journal 79
lation, a secondary endpoint, was also improved with atezolizumab
plus bevacizumab vs sunitinib (median 11.2
vs 8.4 months; HR 0.83, 95% CI 0.70-0.97). Of note, an
independent radiology committee assessed-PFS was also
obtained and differed from the investigator-assessed PFS
in the PD-L1+ population (median 8.9 vs 7.2 months).
ORRs were not significantly different between atezolizumab
plus bevacizumab vs sunitinib in both the PDL1+
(43% vs 35%) and ITT populations (37% vs 33%).
However, complete responses (CRs) were move common
with atezolizumab plus bevacizumab vs sunitinib in both
the PD-L1+ (9% vs 4%) and ITT populations (5% vs 2%).
At the first interim analysis, OS data was immature, but
suggestive of an encouraging trend favoring atezolizumab
plus bevacizumab in both the PD-L1+ (HR 0.81, 95% CI
0.63-1.03; p = 0.09) and ITT (HR 0.68, 95% CI 0.46-1.00)
populations. Follow-up of OS results will likely be crucial
in the regulatory review of this regimen.
Atezolizumab plus bevacizumab was relatively welltolerated
compared to sunitinib, with grade 3-4 treatment
related AEs occurring in 40% vs 54% of patients,
respectively. The most common grade 3-4 treatment-related
AEs with atezolizumab plus bevacizumab included
hypertension, proteinuria, and asthenia. Corticosteroids
were given to 16% of patients treated with atezolizumab
plus bevacizumab within 30 days of an AE of special interest.
Treatment-related AEs led to therapy discontinuation
in 12% of patients receiving atezolizumab plus
bevacizumab and 8% of patients receiving sunitinib. Finally,
quality-of life evaluations demonstrated significantly
prolonged time to symptom interference with
activities of daily living in patients treated with atezolizumab
plus bevacizumab as compared to those treated
with sunitinib (HR 0.56, 95% CI 0.46-0.68).
Interestingly, earlier evaluation of the combination of
atezolizumab plus bevacizumab in a small number of
mRCC patients demonstrated molecular and cellular
changes consistent with the pre-clinical data that antiangiogenic
therapy modulates the anti-tumor immune
response.28 Tumor and blood based analyses after a 2 to
3 week lead-in of bevacizumab monotherapy demonstrated
decreases in vascular markers as well as increases
in gene signatures associated with T-helper 1 chemokines
involved in lymphocyte trafficking, tumor MHC-I protein
expression, and infiltration of tumor-specific T-cell
clones. The subsequent addition of atezolizumab to bevacizumab
resulted in anti-tumor activity that was associated
with further increases in intra-tumor CD8+ T cells
and the number of unique T-cell clones within the tumor
microenvironment. Furthermore, the randomized phase
II IMmotion150 study (NCT01984242) identified gene
signatures associated with T-effector and myeloid inflammatory
responses in pre-treatment tumor samples to
significantly predict for response to atezolizumab plus bevacizumab
vs atezolizumab monotherapy vs sunitinib.29
The early studies on atezolizumab plus bevacizumab have
provided thought-provoking insights into the pharmacodynamics
of immune checkpoint and anti-angiogenic
therapy that warrant further investigation.
Pembrolizumab plus Axitinib
While studies combining VEGFR-targeted TKIs with PD-
1/PD-L1 checkpoint inhibitors have shown clinical activity
in mRCC, some combinations have been limited
by unacceptable toxicity, including severe liver function
abnormalities and fatigue.26,27 Many of the toxicities are
thought to be related to the augmentation of off-target
effects of multi-targeted TKIs by immune checkpoint inhibitors.
In response, one approach has been to use a
more specific inhibitor of VEGFR that may be better tolerated
in combination with an anti-PD-1 drug. This was
the rationale underlying a phase Ib trial of axitinib—a potent,
selective inhibitor of VEGFR 1-3—in combination
with pembrolizumab in treatment-naïve patients (n=52)
with advanced ccRCC.22
The combination of pembrolizumab plus axitinib