Kidney Cancer Journal 91
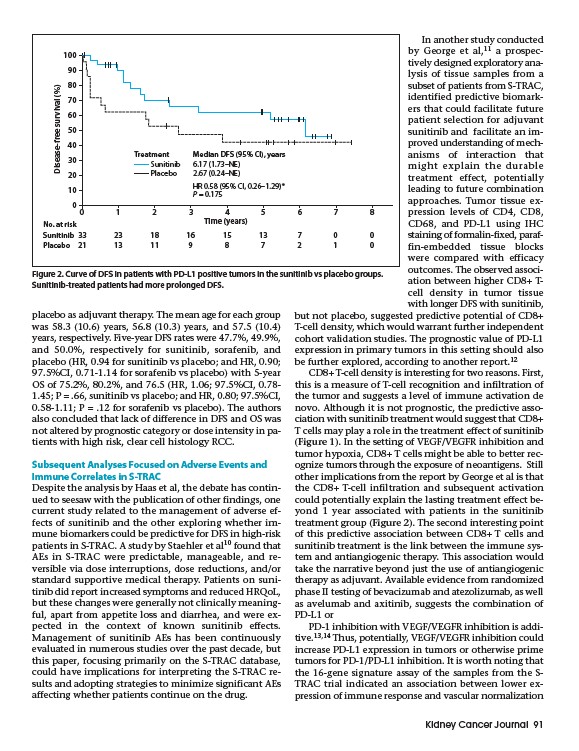
Treatment Median DFS (95% CI), years
Sunitinib
Placebo
6.17 (1.73–NE)
2.67 (0.24–NE)
HR 0.58 (95% CI, 0.26–1.29)*
P = 0.175
placebo as adjuvant therapy. The mean age for each group
was 58.3 (10.6) years, 56.8 (10.3) years, and 57.5 (10.4)
years, respectively. Five-year DFS rates were 47.7%, 49.9%,
and 50.0%, respectively for sunitinib, sorafenib, and
placebo (HR, 0.94 for sunitinib vs placebo; and HR, 0.90;
97.5%CI, 0.71-1.14 for sorafenib vs placebo) with 5-year
OS of 75.2%, 80.2%, and 76.5 (HR, 1.06; 97.5%CI, 0.78-
1.45; P = .66, sunitinib vs placebo; and HR, 0.80; 97.5%CI,
0.58-1.11; P = .12 for sorafenib vs placebo). The authors
also concluded that lack of difference in DFS and OS was
not altered by prognostic category or dose intensity in patients
with high risk, clear cell histology RCC.
Subsequent Analyses Focused on Adverse Events and
Immune Correlates in S-TRAC
Despite the analysis by Haas et al, the debate has continued
to seesaw with the publication of other findings, one
current study related to the management of adverse effects
of sunitinib and the other exploring whether immune
biomarkers could be predictive for DFS in high-risk
patients in S-TRAC. A study by Staehler et al10 found that
AEs in S-TRAC were predictable, manageable, and reversible
via dose interruptions, dose reductions, and/or
standard supportive medical therapy. Patients on sunitinib
did report increased symptoms and reduced HRQoL,
but these changes were generally not clinically meaningful,
apart from appetite loss and diarrhea, and were expected
in the context of known sunitinib effects.
Management of sunitinib AEs has been continuously
evaluated in numerous studies over the past decade, but
this paper, focusing primarily on the S-TRAC database,
could have implications for interpreting the S-TRAC results
and adopting strategies to minimize significant AEs
affecting whether patients continue on the drug.
In another study conducted
by George et al,11 a prospectively
designed exploratory analysis
of tissue samples from a
subset of patients from S-TRAC,
identified predictive biomarkers
that could facilitate future
patient selection for adjuvant
sunitinib and facilitate an improved
understanding of mechanisms
of interaction that
might explain the durable
treatment effect, potentially
leading to future combination
approaches. Tumor tissue expression
levels of CD4, CD8,
CD68, and PD-L1 using IHC
staining of formalin-fixed, paraffin
embedded tissue blocks
were compared with efficacy
outcomes. The observed association
between higher CD8+ Tcell
density in tumor tissue
with longer DFS with sunitinib,
but not placebo, suggested predictive potential of CD8+
T-cell density, which would warrant further independent
cohort validation studies. The prognostic value of PD-L1
expression in primary tumors in this setting should also
be further explored, according to another report.12
CD8+ T-cell density is interesting for two reasons. First,
this is a measure of T-cell recognition and infiltration of
the tumor and suggests a level of immune activation de
novo. Although it is not prognostic, the predictive association
with sunitinib treatment would suggest that CD8+
T cells may play a role in the treatment effect of sunitinib
(Figure 1). In the setting of VEGF/VEGFR inhibition and
tumor hypoxia, CD8+ T cells might be able to better recognize
tumors through the exposure of neoantigens. Still
other implications from the report by George et al is that
the CD8+ T-cell infiltration and subsequent activation
could potentially explain the lasting treatment effect beyond
1 year associated with patients in the sunitinib
treatment group (Figure 2). The second interesting point
of this predictive association between CD8+ T cells and
sunitinib treatment is the link between the immune system
and antiangiogenic therapy. This association would
take the narrative beyond just the use of antiangiogenic
therapy as adjuvant. Available evidence from randomized
phase II testing of bevacizumab and atezolizumab, as well
as avelumab and axitinib, suggests the combination of
PD-L1 or
PD-1 inhibition with VEGF/VEGFR inhibition is additive.
13,14 Thus, potentially, VEGF/VEGFR inhibition could
increase PD-L1 expression in tumors or otherwise prime
tumors for PD-1/PD-L1 inhibition. It is worth noting that
the 16-gene signature assay of the samples from the STRAC
trial indicated an association between lower expression
of immune response and vascular normalization
100
90
80
70
60
50
40
30
20
10
0
0 1 2 3 4 5 6 7 8
Time (years)
Disease-free survival (%)
No. at risk
Sunitinib 33 23 18 16 15 13 7 0 0
Placebo 21 13 11 9 8 7 2 1 0
Figure 2. Curve of DFS in patients with PD-L1 positive tumors in the sunitinib vs placebo groups.
Sunitinib-treated patients had more prolonged DFS.