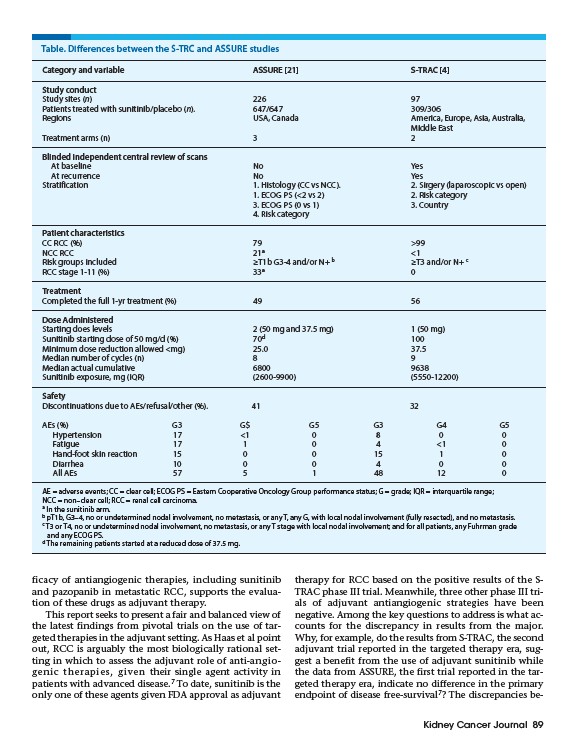
Table. Differences between the S-TRC and ASSURE studies
Category and variable ASSURE 21 S-TRAC 4
Study conduct
Study sites (n) 226 97
Patients treated with sunitinib/placebo (n). 647/647 309/306
Regions USA, Canada America, Europe, Asia, Australia,
Middle East
Treatment arms (n) 3 2
Blinded independent central review of scans
At baseline No Yes
At recurrence No Yes
Stratification 1. Histology (CC vs NCC). 2. Sirgery (laparoscopic vs open)
1. ECOG PS (<2 vs 2) 2. Risk category
3. ECOG PS (0 vs 1) 3. Country
4. Risk category
Patient characteristics
CC RCC (%) 79 >99
NCC RCC 21a <1
Risk groups included ≥T1b G3-4 and/or N+ b ≥T3 and/or N+ c
RCC stage 1-11 (%) 33a 0
Treatment
Completed the full 1-yr treatment (%) 49 56
Dose Administered
Starting does levels 2 (50 mg and 37.5 mg) 1 (50 mg)
Sunitinib starting dose of 50 mg/d (%) 70d 100
Minimum dose reduction allowed <mg) 25.0 37.5
Median number of cycles (n) 8 9
Median actual cumulative 6800 9638
Sunitinib exposure, mg (IQR) (2600-9900) (5550-12200)
Safety
Discontinuations due to AEs/refusal/other (%). 41 32
AEs (%) G3 G$ G5 G3 G4 G5
Hypertension 17 <1 0 8 0 0
Fatigue 17 1 0 4 <1 0
Hand-foot skin reaction 15 0 0 15 1 0
Diarrhea 10 0 0 4 0 0
All AEs 57 5 1 48 12 0
AE = adverse events; CC = clear cell; ECOG PS = Eastern Cooperative Oncology Group performance status; G = grade; IQR = interquartile range;
NCC = non–clear cell; RCC = renal cell carcinoma.
a In the sunitinib arm.
b pT1b, G3–4, no or undetermined nodal involvement, no metastasis, or any T, any G, with local nodal involvement (fully resected), and no metastasis.
c T3 or T4, no or undetermined nodal involvement, no metastasis, or any T stage with local nodal involvement; and for all patients, any Fuhrman grade
and any ECOG PS.
d The remaining patients started at a reduced dose of 37.5 mg.
Kidney Cancer Journal 89
ficacy of antiangiogenic therapies, including sunitinib
and pazopanib in metastatic RCC, supports the evaluation
of these drugs as adjuvant therapy.
This report seeks to present a fair and balanced view of
the latest findings from pivotal trials on the use of targeted
therapies in the adjuvant setting. As Haas et al point
out, RCC is arguably the most biologically rational setting
in which to assess the adjuvant role of anti-angiogenic
therapies, given their single agent activity in
patients with advanced disease.7 To date, sunitinib is the
only one of these agents given FDA approval as adjuvant
therapy for RCC based on the positive results of the STRAC
phase III trial. Meanwhile, three other phase III trials
of adjuvant antiangiogenic strategies have been
negative. Among the key questions to address is what accounts
for the discrepancy in results from the major.
Why, for example, do the results from S-TRAC, the second
adjuvant trial reported in the targeted therapy era, suggest
a benefit from the use of adjuvant sunitinib while
the data from ASSURE, the first trial reported in the targeted
therapy era, indicate no difference in the primary
endpoint of disease free-survival7? The discrepancies be-