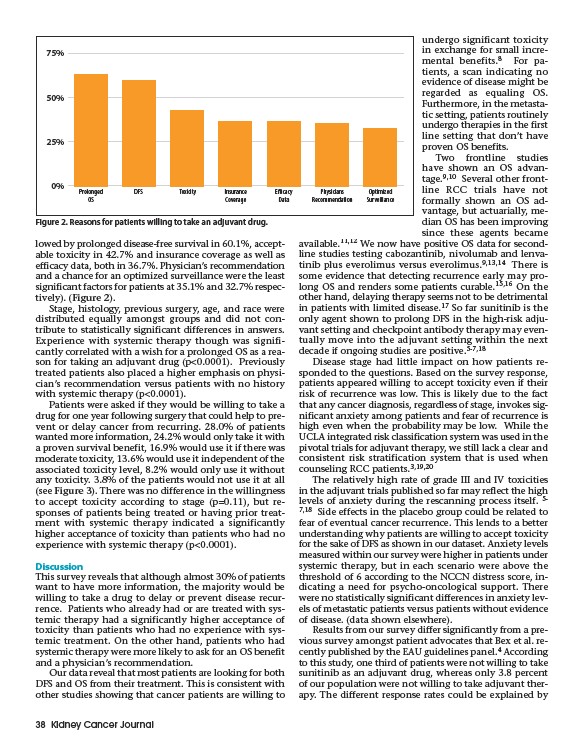
lowed by prolonged disease-free survival in 60.1%, acceptable
toxicity in 42.7% and insurance coverage as well as
efficacy data, both in 36.7%. Physician’s recommendation
and a chance for an optimized surveillance were the least
significant factors for patients at 35.1% and 32.7% respectively).
(Figure 2).
Stage, histology, previous surgery, age, and race were
distributed equally amongst groups and did not contribute
to statistically significant differences in answers.
Experience with systemic therapy though was significantly
correlated with a wish for a prolonged OS as a reason
for taking an adjuvant drug (p<0.0001). Previously
treated patients also placed a higher emphasis on physician’s
recommendation versus patients with no history
with systemic therapy (p<0.0001).
Patients were asked if they would be willing to take a
drug for one year following surgery that could help to prevent
or delay cancer from recurring. 28.0% of patients
wanted more information, 24.2% would only take it with
a proven survival benefit, 16.9% would use it if there was
moderate toxicity, 13.6% would use it independent of the
associated toxicity level, 8.2% would only use it without
any toxicity. 3.8% of the patients would not use it at all
(see Figure 3). There was no difference in the willingness
to accept toxicity according to stage (p=0.11), but responses
of patients being treated or having prior treatment
with systemic therapy indicated a significantly
higher acceptance of toxicity than patients who had no
experience with systemic therapy (p<0.0001).
Discussion
This survey reveals that although almost 30% of patients
want to have more information, the majority would be
willing to take a drug to delay or prevent disease recurrence.
Patients who already had or are treated with systemic
therapy had a significantly higher acceptance of
toxicity than patients who had no experience with systemic
treatment. On the other hand, patients who had
systemic therapy were more likely to ask for an OS benefit
and a physician’s recommendation.
Our data reveal that most patients are looking for both
DFS and OS from their treatment. This is consistent with
other studies showing that cancer patients are willing to
38 Kidney Cancer Journal
undergo significant toxicity
in exchange for small incremental
benefits.8 For patients,
a scan indicating no
evidence of disease might be
regarded as equaling OS.
Furthermore, in the metastatic
setting, patients routinely
undergo therapies in the first
line setting that don’t have
proven OS benefits.
Two frontline studies
have shown an OS advantage.
9,10 Several other frontline
RCC trials have not
formally shown an OS advantage,
but actuarially, median
OS has been improving
since these agents became
available.11,12 We now have positive OS data for secondline
studies testing cabozantinib, nivolumab and lenvatinib
plus everolimus versus everolimus.9,13,14 There is
some evidence that detecting recurrence early may prolong
OS and renders some patients curable.15,16 On the
other hand, delaying therapy seems not to be detrimental
in patients with limited disease.17 So far sunitinib is the
only agent shown to prolong DFS in the high-risk adjuvant
setting and checkpoint antibody therapy may eventually
move into the adjuvant setting within the next
decade if ongoing studies are positive.5-7,18
Disease stage had little impact on how patients responded
to the questions. Based on the survey response,
patients appeared willing to accept toxicity even if their
risk of recurrence was low. This is likely due to the fact
that any cancer diagnosis, regardless of stage, invokes significant
anxiety among patients and fear of recurrence is
high even when the probability may be low. While the
UCLA integrated risk classification system was used in the
pivotal trials for adjuvant therapy, we still lack a clear and
consistent risk stratification system that is used when
counseling RCC patients.3,19,20
The relatively high rate of grade III and IV toxicities
in the adjuvant trials published so far may reflect the high
levels of anxiety during the rescanning process itself. 5-
7,18 Side effects in the placebo group could be related to
fear of eventual cancer recurrence. This lends to a better
understanding why patients are willing to accept toxicity
for the sake of DFS as shown in our dataset. Anxiety levels
measured within our survey were higher in patients under
systemic therapy, but in each scenario were above the
threshold of 6 according to the NCCN distress score, indicating
a need for psycho-oncological support. There
were no statistically significant differences in anxiety levels
of metastatic patients versus patients without evidence
of disease. (data shown elsewhere).
Results from our survey differ significantly from a previous
survey amongst patient advocates that Bex et al. recently
published by the EAU guidelines panel.4 According
to this study, one third of patients were not willing to take
sunitinib as an adjuvant drug, whereas only 3.8 percent
of our population were not willing to take adjuvant therapy.
The different response rates could be explained by
Prolonged
OS
DFS Toxicity Insurance
Coverage
Efficacy
Data
Physicians
Recommendation
Optimized
Surveillance
75%
50%
25%
0%
Figure 2. Reasons for patients willing to take an adjuvant drug.