Pazopanib (2009)
Bevacizumab+IFNα
Everolimus (2009)
Temsinolimus (2007)
Sunitinib (2006)
Sorafenib (2005)
French Model (1L)
(2002)
the GRCC model. The two additional prognostic factors
that were found to improve the predictive ability of the
model are: tumor burden and time to first to second-line
treatment. As opposed to IMDC and MSKCC models, 4
risk groups are defined in the GRCC model: good risk (0
risk factor), intermediate risk (1 to 2 risk factors), low poor
risk (3 to 4 risks factors), and high poor risk (more than 5
risk factors). The median OS of these groups are: 49.5
(26.3-NE), 26.6 (23.1 to 33.1), 12.5 (10.0 to 16.6) and 4.3
(3.9 to 7.2) months respectively.
External validation of the GRCC model and its comparison
with the IMDC and three-factor MSKCC models
was done using a cohort of patients from the INTROSECT
and AXIS trials.17 Overall, the GRCC model has the best
performance in the discovery and validation dataset compared
to the IMDC and MSKCC models. Using the likelihood
ratio, GRCC is found to improve the fit of data in
comparison to the MSKCC model and the IMDC model
44 Kidney Cancer Journal
(2009)
Axitinib+Pembrolizumab
(2019)
Nivolumab+Ipilimumab
(2018)
Cabozantinib (2016)
Lenvatinib+Everolimus
(2016)
Axitinib
(2012) Nivolumab (2015)
in the discovery cohort. Comparing the association of the
prognostic model risk groups and the outcomes with R2
statistics, GRCC strata appears to be more strongly associated
with outcomes than the IMDC and MSKCC model
risk groups, in the discovery and validation cohorts. However,
in the validation cohort, concordance statistics
suggests that GRCC might have similar discriminant ability
to the IMDC model. Using patients solely from the
INTORSECT and AXIS trials, that had received either bevacizumab
or sunitinib as first-line therapy, to validate
the GRCC model might raise concerns regarding the generalizability
of the GRCC model to the real world population.
1,2,3
A paucity of prognostic models exists in the subsequent
lines of treatment. Despite being developed in the
first-line setting, evidence also supports the prognostic
ability of the IMDC model in third- and fourth-line treatment.
12,18 The use of the IMDC model might have im-
Cytokine
Anti-VEGF TKI
mTOR Inhibitor
Anti-VEGF mAB
ICP
Drug Combinations
Undergoing FDA Review
Cytokine Era
TTx Era
ICP Era
Legend
IL-2 and IFNα
HD IL-2
(1992)
1980 1990 2000 2010 2020
MSKCC Model (1L)
(2002)
CCF Model (1L)
(2007)
IMDC Model (1L)
(2009)
IMDC Model (2L)
(2015)
IMDC Model (1, 2L)
Javelin Renal 101,
Keynote-426 and
Nivoren trials
(2019)
GRCC Model (2L)
(2019)
IKCWG Model (1L)
(2011)
Three-Factors MSKCC Model (2L)
(2002)
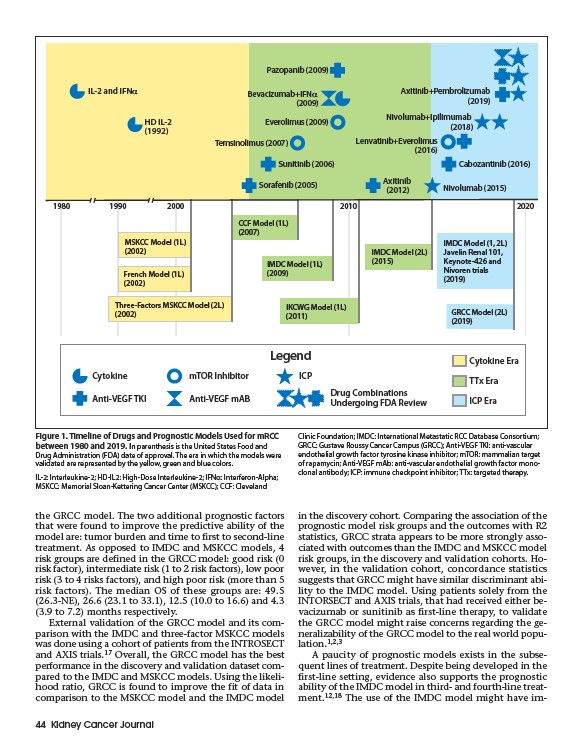
Figure 1. Timeline of Drugs and Prognostic Models Used for mRCC
between 1980 and 2019. In parenthesis is the United States Food and
Drug Administration (FDA) date of approval. The era in which the models were
validated are represented by the yellow, green and blue colors.
IL-2: Interleukine-2; HD-IL2: High-Dose Interleukine-2; IFNa: Interferon-Alpha;
MSKCC: Memorial Sloan-Kettering Cancer Center (MSKCC); CCF: Cleveland
Clinic Foundation; IMDC: International Metastatic RCC Database Consortium;
GRCC: Gustave Roussy Cancer Campus (GRCC); Anti-VEGF TKI: anti-vascular
endothelial growth factor tyrosine kinase inhibitor; mTOR: mammalian target
of rapamycin; Anti-VEGF mAb: anti-vascular endothelial growth factor monoclonal
antibody; ICP: immune checkpoint inhibitor; TTx: targeted therapy.