Kidney Cancer Journal 49
clear cell histology, sarcomatoid differentiation, T2-4
disease, tumor dimension >10 cm, and N+ disease. These
risk factors were significantly associated with BM at RCC
diagnosis. This report found that patients with BM were
more likely to succumb to any death than those without
BM at diagnosis (median overall survival: 6.4 months vs
not reached) based on the Surveillance, Epidemiology,
and End Results (SEER) database. Although their model
needs further testing, the authors suggest that the real incidence
of BM at RCC diagnosis is likely underestimated
given that the observed rate likely reflects patients who
presented with symptoms.
Our review seeks to address this issue by focusing on
clinical trials where RCC patients with BM were not excluded.
We also address key areas related to treatment of
BM in RCC, including the mechanisms of why some
agents presumably would be effective in this setting, the
activity of agents studied so far (tyrosine kinase inhibitors
and immunotherapies) and issues related to penetrance
of the CNS, and novel targets.
Mechanisms Determining Response to therapy
The underestimation of the incidence of BM and the poor
prognostic implications of BM in RCC render it imperative
to further evaluate what treatments could make a difference
and how an awareness of the various mechanisms
of these agents can help to inform management strategies.
Evidence has focused on a broad spectrum of agents,
beginning with the VEGFR tyrosine kinase inhibitors. The
TKIs that are FDA-approved for the treatment of mRCC
include sunitinib, pazopanib, sorafenib, axitinib, cabozantinib
and lenvatinib. Despite their efficacy as standard
therapies for patients with metastatic clear cell RCC and
extracranial disease, the CNS response rates in RCC with
BM have been modest, suggesting that the antiangiogenic
mechanism of action of TKIs does not produce the anticipated
significant efficacy. Overall, the evidence that
TKIs may have activity in the brain is disappointing.
There was early evidence of a promising effect. For example,
retrospective analysis of a phase 3 trial randomizing
patients between sorafenib and placebo found lower
crude rates of BM in the group receiving the drug.5 Similarly,
another report found sunitinib and sorafenib to be
protective with regard to BM development.6
More recent studies, however, tend to conclude otherwise,
indicating that the impact of these agents on outcomes
in patients with existing BM is limited. A
retrospective review by Verma et al7 suggests that these
agents may provide a survival benefit in patients with
BM, notably in those who are TKI naïve. Yet the definitive
benefit of these agents in this population was unclear as
they did not observe any statistically significant improvement
either in rate of local control or distant brain metastasis
free survival. Rather, these TKIs are likely to have
only a marginal benefit in the management of BM from
RCC as any survival benefit is likely due to improved control
of systemic disease rather than a direct impact on the
brain.
Although the experience with sunitinib and sorafenib
in this setting is disappointing, more favorable results
with another TKI suggests how its mechanism of action
could be associated with significantly improved outcomes.
A growing list of studies evaluating the use of cabozantinib
delineate why this VEGF TKI may offer a better
option to other agents in its class, largely due to a mechanism
that differentiates this TKI from sunitinib and sorafenib.
One of the problems in evaluating the efficacy of
targeted therapies in RCC patients with BM is that this
group has historically been excluded from most prospective
clinical trials. Consider, for example, the METEOR
and CABOSUN trials that allowed treatment of
mRCC patients having BM with cabozantinib; however
this subset was underrepresented (<1%) in METEOR and
not reported in CABOSUN.8,9 Our case report10 nevertheless
pursued the hypothesis advocating the use of cabozantinib
in mRCC patients with BM because the drug
targets MET. The rationale is based on previous findings
that MET expression was observed in 35% of BM compared
to 0% of primary RCC tumors.11
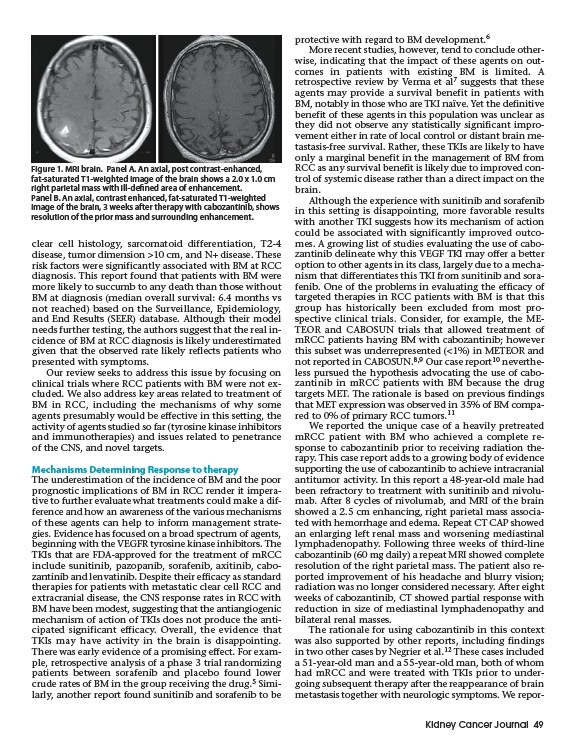
We reported the unique case of a heavily pretreated
mRCC patient with BM who achieved a complete response
to cabozantinib prior to receiving radiation therapy.
This case report adds to a growing body of evidence
supporting the use of cabozantinib to achieve intracranial
antitumor activity. In this report a 48-year-old male had
been refractory to treatment with sunitinib and nivolumab.
After 8 cycles of nivolumab, and MRI of the brain
showed a 2.5 cm enhancing, right parietal mass associated
with hemorrhage and edema. Repeat CT CAP showed
an enlarging left renal mass and worsening mediastinal
lymphadenopathy. Following three weeks of third-line
cabozantinib (60 mg daily) a repeat MRI showed complete
resolution of the right parietal mass. The patient also reported
improvement of his headache and blurry vision;
radiation was no longer considered necessary. After eight
weeks of cabozantinib, CT showed partial response with
reduction in size of mediastinal lymphadenopathy and
bilateral renal masses.
The rationale for using cabozantinib in this context
was also supported by other reports, including findings
in two other cases by Negrier et al.12 These cases included
a 51-year-old man and a 55-year-old man, both of whom
had mRCC and were treated with TKIs prior to undergoing
subsequent therapy after the reappearance of brain
metastasis together with neurologic symptoms. We repor-
Figure 1. MRI brain. Panel A. An axial, post contrast-enhanced,
fat-saturated T1-weighted image of the brain shows a 2.0 x 1.0 cm
right parietal mass with ill-defined area of enhancement.
Panel B. An axial, contrast enhanced, fat-saturated T1-weighted
image of the brain, 3 weeks after therapy with cabozantinib, shows
resolution of the prior mass and surrounding enhancement.