More information needed
Use it only if it prolongs survival
Use it if there was moderate toxicity
Use it no matter what toxicity level
Use it only if there was no toxicity
Do not use it
4%
30%
26%
14%
18%
9%
the fact that our question was designed specifically to
eliminate bias toward or against therapy decisions. In
contrast, the question posed in the previous survey, “After
surgery for kidney cancer, if your doctor told that you are
at high risk of recurrence (spread), would you consider
taking a drug for one year in the hope you could delay
the onset of recurrence even if your OS was not improved?”
included specific contextualization of the question
not included in the current survey. Given the ex-
planatory nature of the question, it can be debated
whether the patient’s perspective was fully accurately captured
with that approach.21-25 The current questions were
designed to estimate what matters to patients, whether
the questionnaire recently published by the EAU guidelines
panel was investigating the perception of boundaries
for acceptance of adjuvant therapy based on
estimated results of the S-TRAC trial. Slight differences in
questioning patients might lead to different interpretation
of the underlying perspective have to be taken into
consideration when counseling and analyzing patients
needs. S-TRAC was designed with a primary endpoint of
PFS, and OS was a secondary endpoint. The FDA approval
was given based on the improvement in PFS, with
a hazard of 0.76 with an absolute improvement in 5 year
DFS of 8%. These data are in line with other approved
adjuvant indications, and as no additional analyses are
planned, patients should be provided the necessary context
to interpret the lack of an OS signal in the S-TRAC
study.
Although our dataset was open to the entire patient
population and included all histologies and stages it
might be biased by the higher rate of females and a lower
median age compared to the usual epidemiology of RCC.
We suspect that the higher participation rate among these
populations was due to an increased willingness to engage
in social media and on-line patient support groups;
however, highly possible these variances did not significantly
impact the overall data. Additionally, our study
demonstrates that questionnaires/surveys distributed by
a patient advocacy organization on international patient
fora can successfully solicit responses from a relatively
large cohort of patients and can serve as model for future
research on patient preference, understanding and decision
making.
Conclusion
Patients opinions seem to differ from physician’s expectations.
Regardless of stage, patients perceive themselves
as being at high risk of recurrence and are willing to undergo
adjuvant treatment regardless of toxicity. In addition,
patients don’t differentiate between DFS and OS.
Given that anxiety across risk groups is high and that
the majority of patients are willing to accept toxicity to
prevent recurrence, it’s critical that if adjuvant treatment
is approved, appropriate patient education tools are developed
so patients can effectively understand and make
informed decisions.
References
1. Global Burden of Disease Cancer C, Fitzmaurice C, Dicker D, et al.
The Global Burden of Cancer 2013. JAMA Oncol. 2015;1(4):505-527.
2. Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, et al. Cancer incidence
and mortality patterns in Europe: estimates for 40 countries in 2012.
Eur J Cancer. 2013;49(6):1374-1403.
3. Lam JS, Shvarts O, Leppert JT, Pantuck AJ, Figlin RA, Belldegrun AS.
Postoperative surveillance protocol for patients with localized and locally
advanced renal cell carcinoma based on a validated prognostic
nomogram and risk group stratification system. J Urol. 2005;174(2):466-
472; discussion 472; quiz 801.
4. Bex A, Albiges L, Ljungberg B, et al. Updated European Association
of Urology Guidelines Regarding Adjuvant Therapy for Renal Cell Carcinoma.
Eur Urol. 2017;71(5):719-722.
5. Ravaud A, Motzer RJ, Pandha HS, et al. Adjuvant Sunitinib in High-
Risk Renal-Cell Carcinoma after Nephrectomy. N Engl J Med. 2016.
6. Haas NB, Manola J, Uzzo RG, et al. Adjuvant sunitinib or sorafenib
for high-risk, non-metastatic renal-cell carcinoma (ECOG-ACRIN
E2805): a double-blind, placebo-controlled, randomised, phase 3 trial.
Lancet. 2016.
7. Motzer R, Haas N, Donskov F, et al. Randomized phase III trial of adjuvant
pazopanib versus placebo after nephrectomy in patients with locally
advanced renal cell carcinoma (RCC) (PROTECT). J Clin Oncol.
2017;35 (suppl. abstr. 4507).
8. Slevin ML, Stubbs L, Plant HJ, et al. Attitudes to chemotherapy: comparing
views of patients with cancer with those of doctors, nurses, and
general public. BMJ. 1990;300(6737):1458-1460.
9. Hudes G, Carducci M, Tomczak P, et al. A phase 3, randomized, 3-
arm study of temsirolimus (TEMSR) or interferon-alpha (IFN) or the
combination of TEMSR + IFN in the treatment of first-line, poor-risk patients
with advanced renal cell carcinoma (adv RCC) (Late breaking Abstract,
ASCO 2006). J Clin Oncol. 2006;24(18S Part II of II):930s, LBA934.
10. Motzer RJ, Tannir NM, McDermott DF, et al. Nivolumab plus Ipilimumab
versus Sunitinib in Advanced Renal-Cell Carcinoma. N Engl J
Med. 2018;378(14):1277-1290.
11. Heng DY, Xie W, Regan MM, et al. Prognostic factors for overall survival
in patients with metastatic renal cell carcinoma treated with vas-
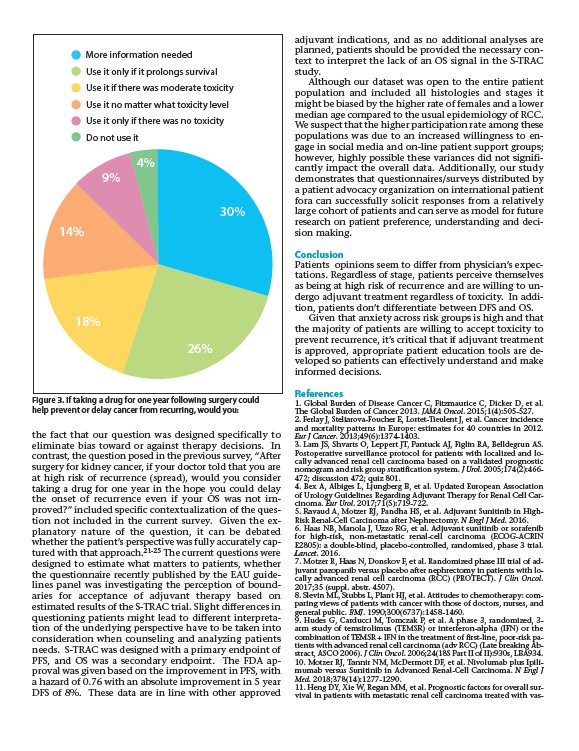
Figure 3. If taking a drug for one year following surgery could
help prevent or delay cancer from recurring, would you: