Kidney Cancer Journal 37
juvant therapy. Given the conflicting data from multiple
trials, significant controversy exists as to whether the clinical
use of sunitinib in the adjuvant setting is justified.
Critics of the S-TRAC data often cite concerns related to
the short duration of DFS improvement, lack of OS trends
and toxicity associated with treatment as reasons to question
clinical use. However, a critically overlooked question
is what factors and wishes drive patients when
making treatment decisions.
Given the fact that adjuvant sunitinib has been recently
approved by the US Food and Drug Administration
(FDA), the purpose of our study was to investigate the patients
perspective on adjuvant therapy in RCC.
Material and Methods
The patient survey was designed by members of the European
Association of Urology (EAU) Renal Cell Carcinoma
Guidelines Panel and KCCure, a U.S. based
non-profit patient advocacy organization, specializing in
research funding for kidney cancer. The survey included
a total of 12 questions that addressed patient’s concerns
and considerations related to adjuvant therapy, as well as
questions about their disease, surveillance regimes and
overall anxiety related to their diagnosis (Figure 1). The
questionnaire was hosted on KCCure’s
website and posted on international
patient forums ad-
dressing approximately 800 patients
between April 1st and June
15th, 2017. Duplicate responses
were eliminated before the data
were analyzed. The same questions
were answered by 19 patient
advocates during an ad-
vocacy organization meeting in
Warsaw, Poland and compared
to the KCCure patients. The
complete survey can be seen in
supplemental material.
Statistical analyses Medians
were calculated with a confidence
interval (CI) of 95% and
an alpha of 0.05 using SPSS statistics
25.0 (IBM Corp., Armonk,
New York, USA). We used Kaplan–
Meier methods to determine
the median duration until
response since diagnosis. Significance
among the different
groups was calculated using
Kruskal-Wallis test with a significance
level of 0.05.
Results
Out of n=653 potential patients
on the webpage n=450 responses
were generated for analysis. Responses
were collected from the
U.S., Canada, South Africa, Great
Britain, Australia, France and
Germany. Median age was 55.6
years (17-82 years) and 56.4% of
the patients were female; 73.6% of the patients had a
nephrectomy as primary therapy, while 22.0% had a partial
nephrectomy. The majority of the patients had clear
cell RCC (76.4%), followed by unclassified RCC (3.9%),
papillary type I or type II RCC (3.6%), chromophobe RCC
(3.6%), translocation RCC (2.0%), collecting duct carcinoma
(0.7%) and renal medullary carcinoma (0.2%), and
9.1% of the patients without knowing their histology.
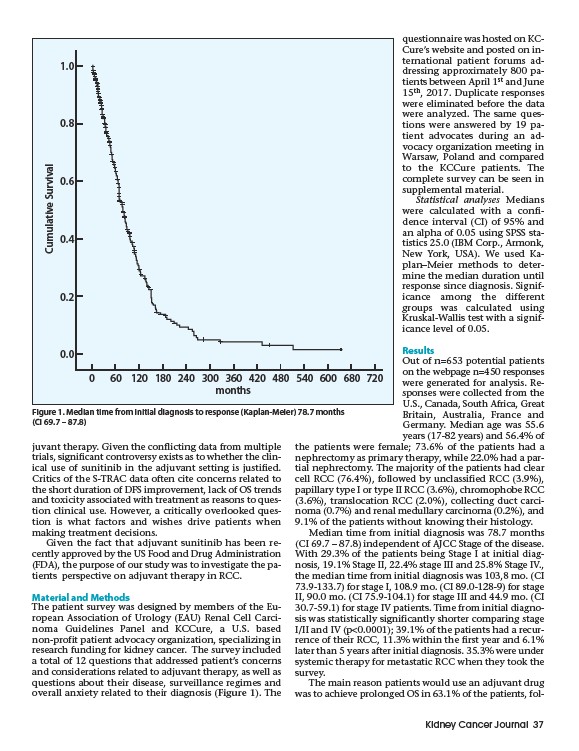
Median time from initial diagnosis was 78.7 months
(CI 69.7 – 87.8) independent of AJCC Stage of the disease.
With 29.3% of the patients being Stage I at initial diagnosis,
19.1% Stage II, 22.4% stage III and 25.8% Stage IV.,
the median time from initial diagnosis was 103,8 mo. (CI
73.9-133.7) for stage I, 108.9 mo. (CI 89.0-128-9) for stage
II, 90.0 mo. (CI 75.9-104.1) for stage III and 44.9 mo. (CI
30.7-59.1) for stage IV patients. Time from initial diagnosis
was statistically significantly shorter comparing stage
I/II and IV (p<0.0001); 39.1% of the patients had a recurrence
of their RCC, 11.3% within the first year and 6.1%
later than 5 years after initial diagnosis. 35.3% were under
systemic therapy for metastatic RCC when they took the
survey.
The main reason patients would use an adjuvant drug
was to achieve prolonged OS in 63.1% of the patients, fol-
1.0
0.8
0.6
0.4
0.2
0.0
0 60 120 180 240 300 360 420 480 540 600 680 720
months
Cumulative Survival
Figure 1. Median time from initial diagnosis to response (Kaplan-Meier) 78.7 months
(CI 69.7 – 87.8)