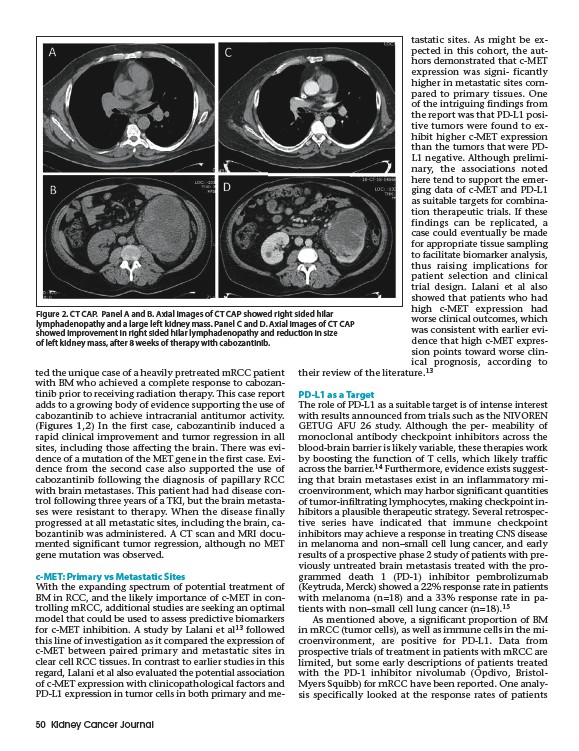
Figure 2. CT CAP. Panel A and B. Axial images of CT CAP showed right sided hilar
lymphadenopathy and a large left kidney mass. Panel C and D. Axial images of CT CAP
showed improvement in right sided hilar lymphadenopathy and reduction in size
of left kidney mass, after 8 weeks of therapy with cabozantinib.
ted the unique case of a heavily pretreated mRCC patient
with BM who achieved a complete response to cabozantinib
prior to receiving radiation therapy. This case report
adds to a growing body of evidence supporting the use of
cabozantinib to achieve intracranial antitumor activity.
(Figures 1,2) In the first case, cabozantinib induced a
rapid clinical improvement and tumor regression in all
sites, including those affecting the brain. There was evidence
of a mutation of the MET gene in the first case. Evidence
from the second case also supported the use of
cabozantinib following the diagnosis of papillary RCC
with brain metastases. This patient had had disease control
following three years of a TKI, but the brain metastases
were resistant to therapy. When the disease finally
progressed at all metastatic sites, including the brain, cabozantinib
was administered. A CT scan and MRI documented
significant tumor regression, although no MET
gene mutation was observed.
c-MET: Primary vs Metastatic Sites
With the expanding spectrum of potential treatment of
BM in RCC, and the likely importance of c-MET in controlling
mRCC, additional studies are seeking an optimal
model that could be used to assess predictive biomarkers
for c-MET inhibition. A study by Lalani et al13 followed
this line of investigation as it compared the expression of
c-MET between paired primary and metastatic sites in
clear cell RCC tissues. In contrast to earlier studies in this
regard, Lalani et al also evaluated the potential association
of c-MET expression with clinicopathological factors and
PD-L1 expression in tumor cells in both primary and metastatic
50 Kidney Cancer Journal
sites. As might be expected
in this cohort, the authors
demonstrated that c-MET
expression was signi- ficantly
higher in metastatic sites compared
to primary tissues. One
of the intriguing findings from
the report was that PD-L1 positive
tumors were found to exhibit
higher c-MET expression
than the tumors that were PDL1
negative. Although preliminary,
the associations noted
here tend to support the emerging
data of c-MET and PD-L1
as suitable targets for combination
therapeutic trials. If these
findings can be replicated, a
case could eventually be made
for appropriate tissue sampling
to facilitate biomarker analysis,
thus raising implications for
patient selection and clinical
trial design. Lalani et al also
showed that patients who had
high c-MET expression had
worse clinical outcomes, which
was consistent with earlier evidence
that high c-MET expression
points toward worse clin-
ical prognosis, according to
their review of the literature.13
PD-L1 as a Target
The role of PD-L1 as a suitable target is of intense interest
with results announced from trials such as the NIVOREN
GETUG AFU 26 study. Although the per- meability of
monoclonal antibody checkpoint inhibitors across the
blood-brain barrier is likely variable, these therapies work
by boosting the function of T cells, which likely traffic
across the barrier.14 Furthermore, evidence exists suggesting
that brain metastases exist in an inflammatory microenvironment,
which may harbor significant quantities
of tumor-infiltrating lymphocytes, making checkpoint inhibitors
a plausible therapeutic strategy. Several retrospective
series have indicated that immune checkpoint
inhibitors may achieve a response in treating CNS disease
in melanoma and non–small cell lung cancer, and early
results of a prospective phase 2 study of patients with previously
untreated brain metastasis treated with the programmed
death 1 (PD-1) inhibitor pembrolizumab
(Keytruda, Merck) showed a 22% response rate in patients
with melanoma (n=18) and a 33% response rate in patients
with non–small cell lung cancer (n=18).15
As mentioned above, a significant proportion of BM
in mRCC (tumor cells), as well as immune cells in the microenvironment,
are positive for PD-L1. Data from
prospective trials of treatment in patients with mRCC are
limited, but some early descriptions of patients treated
with the PD-1 inhibitor nivolumab (Opdivo, Bristol-
Myers Squibb) for mRCC have been reported. One analysis
specifically looked at the response rates of patients