Kidney Cancer Journal 21
Figure 2. IL‐2 plays multiple roles in shaping the immune response (Adapted from Mitra et al.).
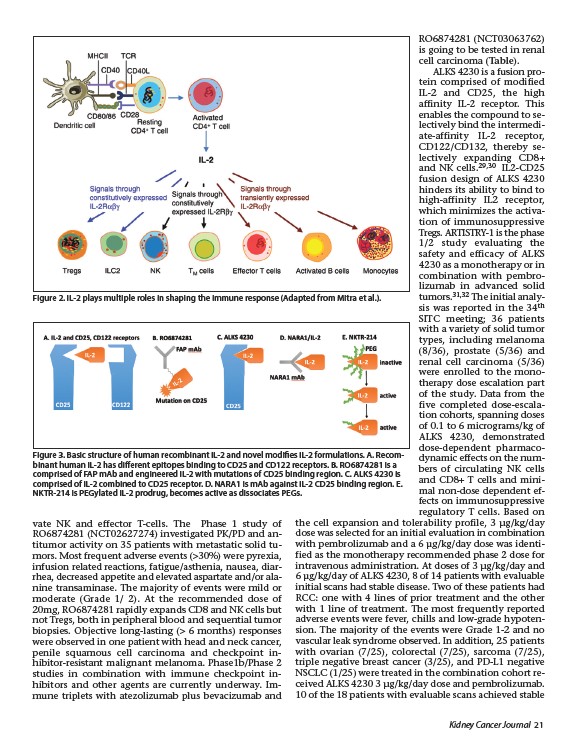
Figure 3. Basic structure of human recombinant IL-2 and novel modifies IL-2 formulations. A. Recombinant
human IL-2 has different epitopes binding to CD25 and CD122 receptors. B. RO6874281 is a
comprised of FAP mAb and engineered IL-2 with mutations of CD25 binding region. C. ALKS 4230 is
comprised of IL-2 combined to CD25 receptor. D. NARA1 is mAb against IL-2 CD25 binding region. E.
NKTR-214 is PEGylated IL-2 prodrug, becomes active as dissociates PEGs.
vate NK and effector T-cells. The Phase 1 study of
RO6874281 (NCT02627274) investigated PK/PD and antitumor
activity on 35 patients with metastatic solid tumors.
Most frequent adverse events (>30%) were pyrexia,
infusion related reactions, fatigue/asthenia, nausea, diarrhea,
decreased appetite and elevated aspartate and/or alanine
transaminase. The majority of events were mild or
moderate (Grade 1/ 2). At the recommended dose of
20mg, RO6874281 rapidly expands CD8 and NK cells but
not Tregs, both in peripheral blood and sequential tumor
biopsies. Objective long-lasting (> 6 months) responses
were observed in one patient with head and neck cancer,
penile squamous cell carcinoma and checkpoint inhibitor
resistant malignant melanoma. Phase1b/Phase 2
studies in combination with immune checkpoint inhibitors
and other agents are currently underway. Immune
triplets with atezolizumab plus bevacizumab and
RO6874281 (NCT03063762)
is going to be tested in renal
cell carcinoma (Table).
ALKS 4230 is a fusion protein
comprised of modified
IL-2 and CD25, the high
affinity IL-2 receptor. This
enables the compound to selectively
bind the intermediate
affinity IL-2 receptor,
CD122/CD132, thereby selectively
expanding CD8+
and NK cells.29,30 IL2-CD25
fusion design of ALKS 4230
hinders its ability to bind to
high-affinity IL2 receptor,
which minimizes the activation
of immunosuppressive
Tregs. ARTISTRY-1 is the phase
1/2 study evaluating the
safety and efficacy of ALKS
4230 as a monotherapy or in
combination with pembrolizumab
in advanced solid
tumors.31,32 The initial analysis
was reported in the 34th
SITC meeting; 36 patients
with a variety of solid tumor
types, including melanoma
(8/36), prostate (5/36) and
renal cell carcinoma (5/36)
were enrolled to the monotherapy
dose escalation part
of the study. Data from the
five completed dose-escalation
cohorts, spanning doses
of 0.1 to 6 micrograms/kg of
ALKS 4230, demonstrated
dose-dependent pharmacodynamic
effects on the numbers
of circulating NK cells
and CD8+ T cells and minimal
non-dose dependent effects
on immunosuppressive
regulatory T cells. Based on
the cell expansion and tolerability profile, 3 μg/kg/day
dose was selected for an initial evaluation in combination
with pembrolizumab and a 6 μg/kg/day dose was identified
as the monotherapy recommended phase 2 dose for
intravenous administration. At doses of 3 μg/kg/day and
6 μg/kg/day of ALKS 4230, 8 of 14 patients with evaluable
initial scans had stable disease. Two of these patients had
RCC: one with 4 lines of prior treatment and the other
with 1 line of treatment. The most frequently reported
adverse events were fever, chills and low-grade hypotension.
The majority of the events were Grade 1-2 and no
vascular leak syndrome observed. In addition, 25 patients
with ovarian (7/25), colorectal (7/25), sarcoma (7/25),
triple negative breast cancer (3/25), and PD-L1 negative
NSCLC (1/25) were treated in the combination cohort received
ALKS 4230 3 μg/kg/day dose and pembrolizumab.
10 of the 18 patients with evaluable scans achieved stable