Kidney Cancer Journal 7
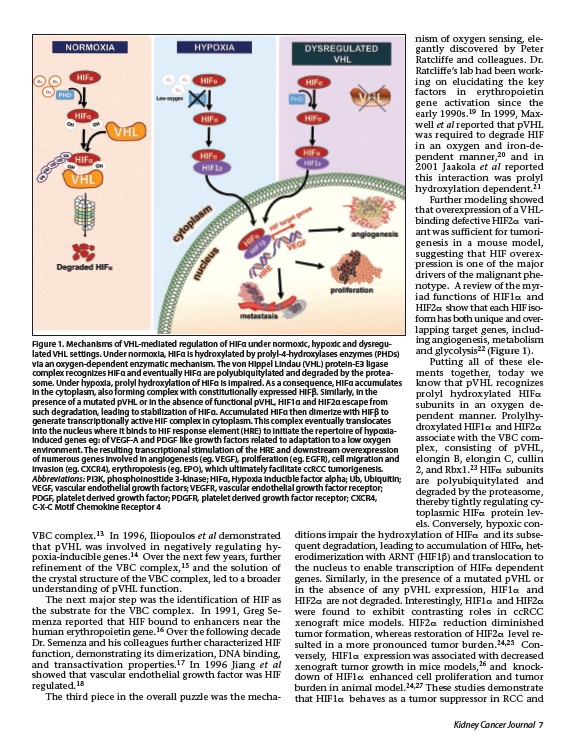
Figure 1. Mechanisms of VHL-mediated regulation of HIFα under normoxic, hypoxic and dysregulated
VHL settings. Under normoxia, HIFα is hydroxylated by prolyl-4-hydroxylases enzymes (PHDs)
via an oxygen-dependent enzymatic mechanism. The von Hippel Lindau (VHL) protein-E3 ligase
complex recognizes HIFα and eventually HIFα are polyubiquitylated and degraded by the proteasome.
Under hypoxia, prolyl hydroxylation of HIFα is impaired. As a consequence, HIFα accumulates
in the cytoplasm, also forming complex with constitutionally expressed HIFβ. Similarly, in the
presence of a mutated pVHL or in the absence of functional pVHL, HIF1α and HIF2α escape from
such degradation, leading to stabilization of HIFα. Accumulated HIFα then dimerize with HIFβ to
generate transcriptionally active HIF complex in cytoplasm. This complex eventually translocates
into the nucleus where it binds to HIF response element (HRE) to initiate the repertoire of hypoxiainduced
genes eg: of VEGF-A and PDGF like growth factors related to adaptation to a low oxygen
environment. The resulting transcriptional stimulation of the HRE and downstream overexpression
of numerous genes involved in angiogenesis (eg. VEGF), proliferation (eg. EGFR), cell migration and
invasion (eg. CXCR4), erythropoiesis (eg. EPO), which ultimately facilitate ccRCC tumorigenesis.
Abbreviations: PI3K, phosphoinositide 3-kinase; HIFα, Hypoxia inducible factor alpha; Ub, Ubiquitin;
VEGF, vascular endothelial growth factors; VEGFR, vascular endothelial growth factor receptor;
PDGF, platelet derived growth factor; PDGFR, platelet derived growth factor receptor; CXCR4,
C-X-C Motif Chemokine Receptor 4
VBC complex.13 In 1996, Iliopoulos et al demonstrated
that pVHL was involved in negatively regulating hypoxia
inducible genes.14 Over the next few years, further
refinement of the VBC complex,15 and the solution of
the crystal structure of the VBC complex, led to a broader
understanding of pVHL function.
The next major step was the identification of HIF as
the substrate for the VBC complex. In 1991, Greg Semenza
reported that HIF bound to enhancers near the
human erythropoietin gene.16 Over the following decade
Dr. Semenza and his colleagues further characterized HIF
function, demonstrating its dimerization, DNA binding,
and transactivation properties.17 In 1996 Jiang et al
showed that vascular endothelial growth factor was HIF
regulated.18
The third piece in the overall puzzle was the mechanism
of oxygen sensing, elegantly
discovered by Peter
Ratcliffe and colleagues. Dr.
Ratcliffe’s lab had been working
on elucidating the key
factors in erythropoietin
gene activation since the
early 1990s.19 In 1999, Maxwell
et al reported that pVHL
was required to degrade HIF
in an oxygen and iron-dependent
manner,20 and in
2001 Jaakola et al reported
this interaction was prolyl
hydroxylation dependent.21
Further modeling showed
that overexpression of a VHL-
binding defective HIF2a variant
was sufficient for tumorigenesis
in a mouse model,
suggesting that HIF overexpression
is one of the major
drivers of the malignant phenotype.
A review of the myriad
functions of HIF1a and
HIF2a show that each HIF isoform
has both unique and over-
lapping target genes, including
angiogenesis, metabolism
and glycolysis22 (Figure 1).
Putting all of these elements
together, today we
know that pVHL recognizes
prolyl hydroxylated HIFa
subunits in an oxygen dependent
manner. Prolylhydroxylated
HIF1a and HIF2a
associate with the VBC complex,
consisting of pVHL,
elongin B, elongin C, cullin
2, and Rbx1.23 HIFa subunits
are polyubiquitylated and
degraded by the proteasome,
thereby tightly regulating cytoplasmic
HIFa protein levels.
Conversely, hypoxic con-
ditions impair the hydroxylation of HIFa and its subsequent
degradation, leading to accumulation of HIFa, heterodimerization
with ARNT (HIF1b) and translocation to
the nucleus to enable transcription of HIFa dependent
genes. Similarly, in the presence of a mutated pVHL or
in the absence of any pVHL expression, HIF1a and
HIF2a are not degraded. Interestingly, HIF1a and HIF2a
were found to exhibit contrasting roles in ccRCC
xenograft mice models. HIF2a reduction diminished
tumor formation, whereas restoration of HIF2a level resulted
in a more pronounced tumor burden.24,25 Conversely,
HIF1a expression was associated with decreased
xenograft tumor growth in mice models,26 and knockdown
of HIF1a enhanced cell proliferation and tumor
burden in animal model.24,27 These studies demonstrate
that HIF1a behaves as a tumor suppressor in RCC and