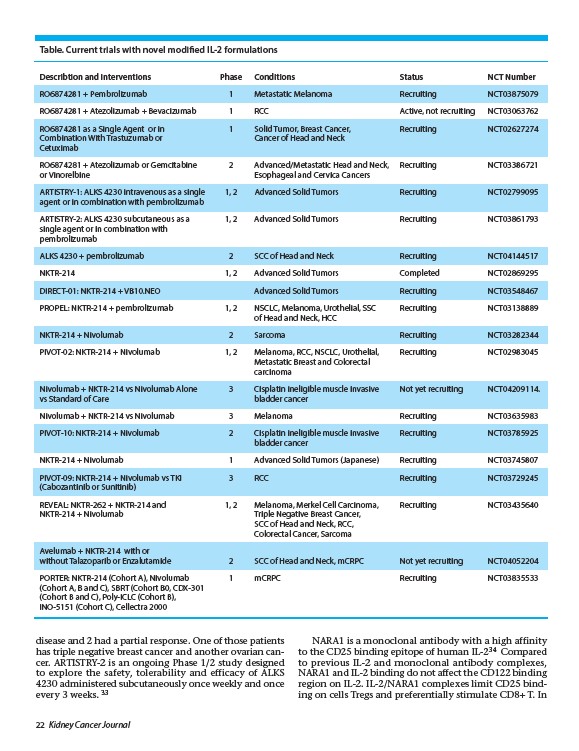
Table. Current trials with novel modified IL-2 formulations
Describtion and Interventions Phase Conditions Status NCT Number
RO6874281 + Pembrolizumab 1 Metastatic Melanoma Recruiting NCT03875079
RO6874281 + Atezolizumab + Bevacizumab 1 RCC Active, not recruiting NCT03063762
RO6874281 as a Single Agent or in 1 Solid Tumor, Breast Cancer, Recruiting NCT02627274
Combination With Trastuzumab or Cancer of Head and Neck
Cetuximab
RO6874281 + Atezolizumab or Gemcitabine 2 Advanced/Metastatic Head and Neck, Recruiting NCT03386721
or Vinorelbine Esophageal and Cervica Cancers
ARTISTRY-1: ALKS 4230 intravenous as a single 1, 2 Advanced Solid Tumors Recruiting NCT02799095
agent or in combination with pembrolizumab
ARTISTRY-2: ALKS 4230 subcutaneous as a 1, 2 Advanced Solid Tumors Recruiting NCT03861793
single agent or in combination with
pembrolizumab
ALKS 4230 + pembrolizumab 2 SCC of Head and Neck Recruiting NCT04144517
NKTR-214 1, 2 Advanced Solid Tumors Completed NCT02869295
DIRECT-01: NKTR-214 + VB10.NEO Advanced Solid Tumors Recruiting NCT03548467
PROPEL: NKTR-214 + pembrolizumab 1, 2 NSCLC, Melanoma, Urothelial, SSC Recruiting NCT03138889
of Head and Neck, HCC
NKTR-214 + Nivolumab 2 Sarcoma Recruiting NCT03282344
PIVOT-02: NKTR-214 + Nivolumab 1, 2 Melanoma, RCC, NSCLC, Urothelial, Recruiting NCT02983045
Metastatic Breast and Colorectal
carcinoma
Nivolumab + NKTR-214 vs Nivolumab Alone 3 Cisplatin ineligible muscle invasive Not yet recruiting NCT04209114.
vs Standard of Care bladder cancer
Nivolumab + NKTR-214 vs Nivolumab 3 Melanoma Recruiting NCT03635983
PIVOT-10: NKTR-214 + Nivolumab 2 Cisplatin ineligible muscle invasive Recruiting NCT03785925
bladder cancer
NKTR-214 + Nivolumab 1 Advanced Solid Tumors (Japanese) Recruiting NCT03745807
PIVOT-09: NKTR-214 + Nivolumab vs TKI 3 RCC Recruiting NCT03729245
(Cabozantinib or Sunitinib)
REVEAL: NKTR-262 + NKTR-214 and 1, 2 Melanoma, Merkel Cell Carcinoma, Recruiting NCT03435640
NKTR-214 + Nivolumab Triple Negative Breast Cancer,
SCC of Head and Neck, RCC,
Colorectal Cancer, Sarcoma
Avelumab + NKTR-214 with or
without Talazoparib or Enzalutamide 2 SCC of Head and Neck, mCRPC Not yet recruiting NCT04052204
PORTER: NKTR-214 (Cohort A), Nivolumab 1 mCRPC Recruiting NCT03835533
(Cohort A, B and C), SBRT (Cohort B0, CDX-301
(Cohort B and C), Poly-ICLC (Cohort B),
INO-5151 (Cohort C), Cellectra 2000
disease and 2 had a partial response. One of those patients
has triple negative breast cancer and another ovarian cancer.
ARTISTRY-2 is an ongoing Phase 1/2 study designed
to explore the safety, tolerability and efficacy of ALKS
4230 administered subcutaneously once weekly and once
every 3 weeks. 33
22 Kidney Cancer Journal
NARA1 is a monoclonal antibody with a high affinity
to the CD25 binding epitope of human IL-234 Compared
to previous IL-2 and monoclonal antibody complexes,
NARA1 and IL-2 binding do not affect the CD122 binding
region on IL-2. IL-2/NARA1 complexes limit CD25 binding
on cells Tregs and preferentially stimulate CD8+ T. In